Bottles Now Have Embedded RFID
A patented technology fully encapsulates Radio Frequency Identification (RFID) components between injection molded discs and then embeds that assembly into an injection-blow molded bottle.
A patented technology fully encapsulates Radio Frequency Identification (RFID) components between injection molded discs and then embeds that assembly into an injection-blow molded bottle. It is the first commercial application of a plastic container with embedded RFID, rather
than a tag affixed to the outside of a product. “The first RFID-embedded bottles are being produced here in the U.S. for the prescription pharmaceutical market,” says Brian Chisholm, R&D Development Engineer at Rexam Primary Packaging in Perrysburg, Ohio. Rexam is producing RFID-embedded pharmaceutical bottles of 40 cc and larger.
The new technology brings improved traceability of the container compared with barcoded labels. Development of an RFID solution was done to comply with upcoming regulations from the FDA requiring drug distributors to provide full traceability throughout the supply chain, says Chisholm.
TWO PROCESSES AVAILABLE
Rexam actually has more than one approach to producing the RFID bottles. Rexam and the former consumer packaging company Owen-Illinois Healthcare Packaging Inc. were separately working on embedded RFID bottle concepts several years ago. Rexam teamed up with Traxxec Ltd. of the U.K. on RFID developmental projects, while O-I Healthcare worked on its own. When Rexam acquired O-I Healthcare in 2007 it ended up with two in-house solutions at its disposal to embed the disc with the encapsulated RFID tag into the base of the container.
One method (not currently in use), which Rexam has licensed from Traxxec, uses a post-mold operation to ultrasonically weld the disc into a pocket in the base of the bottle. The other approach (developed by O-I Healthcare) embeds the disc directly into the bottle base during blow molding. This is the approach Rexam is now using. “During the blow stage of the bottle, we include the tag in the blow mold and embed it in the base of the container,” says Chisholm. “The approach places the disc at the base of the preform just prior to blowing the bottle,” says Chisholm. Rexam developed the industrial machinery to feed, orient, and place the discs into each blow mold. Rexam uses the same material (HDPE) for the disc and the bottle.
This approach employs a proprietary low-pressure, low-temperature molding technique to encapsulate the RFID components (microchip and antenna) between two previously injection molded discs. This overcomes the key hurdle to implementation of molded-in RFID—protecting the electronic components from the heat and pressure of molding.
The technology also overcomes some of the issues presented by external RFID labels on bottles. “It offers less damage to tags since they are protected by the molded disc, resulting in fewer null reads from tag-to-tag contact on conveyor lines,” says Chisholm. The position of the tag in the bottom of the bottle avoids the need to orient the bottle when passing an RFID reader. Additionally, the bottom location enforces a distance between the tags on adjacent bottles, avoiding interference between tags that can lead to “read failure.” The low-pressure/low-temperature molding approach could also be used for sensors or security devices, Chisholm notes.
Rexam uses the Monza RFID chip and antenna technology developed by Impinj Inc., of Seattle. Rexam assembles the RFID components with the injection molded discs and encapsulates the assembly. Rexam inspects and validates each assembly before it is fed to the blow molding process. The bottles are blow molded on standard machines from Jomar Corp., Pleasantville, N.J., and Uniloy Milacron, Inc., Tecumseh, Mich.
Rexam can mold bottles and test them for leaks and RFID functionality in the production line. The pharmaceutical companies do not have to bother anymore with applying RFID-tag labels and checking them. Rexam does it all, says Chisholm.
Read Next
People 4.0 – How to Get Buy-In from Your Staff for Industry 4.0 Systems
Implementing a production monitoring system as the foundation of a ‘smart factory’ is about integrating people with new technology as much as it is about integrating machines and computers. Here are tips from a company that has gone through the process.
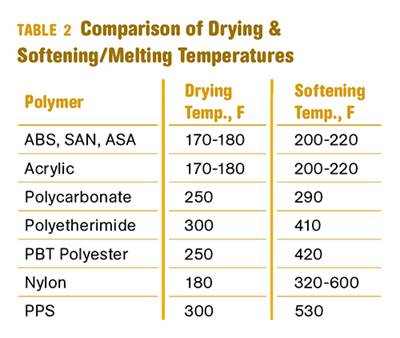
Read MoreWhy (and What) You Need to Dry
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read More







.png;maxWidth=300;quality=90)