Weather Testing: Why You Need It More Than Ever
If you think your product doesn't warrant weather testing, you could be in for a rude surprise. Any time you change a material formulation or use it in a new application you run the risk of uv-induced failure.
Weather testing of plastic parts is becoming a crucial step to head off potential product failures. As OEMs increasingly turn to processors for design and materials selection, the responsibility for in-service weathering failures is also being passed down the supply chain. This is particularly true in automotive plastics. There is often inadequate communication among the OEM, processor, compounder, and additives supplier about the service environment to which the product will be exposed. Misunderstanding how or where a product is to be used is a recipe for product failure.
Manufacturers are under pressure to improve product performance and simultaneously reduce costs. They are turning to new materials and additives as potential solutions. But introducing these unknowns without adequate testing helps explain why the incidence of weathering-related failures is on the rise.
A well-planned weathering test program could identify these risks in advance. Unfortunately, many processors and their customers do not have a good understanding of how to test for weatherability.
Why plastic products fail
There are three main reasons why plastic products fail:
- Bad design, which includes improper material formulation for the end-use environment.
- Using the product outside of design criteria.
- Changes to processes or materials that affect performance.
Failure can mean color and appearance changes such as yellowing, fading, color shift, or gloss loss. For other products, failure is defined in terms of mechanical integrity and physical performance, such as a nylon gear in a washing machine that becomes brittle and cracks.
Customers expect their plastics products to both perform well and look good through the majority of the expected service life. However, it’s up to the customer to define when cosmetic changes add up to product “failure.”
New factors in failures
Plastic materials and products are being introduced into global markets for which they may not have been designed or tested. One of the largest makers of traffic-control products sends specimens for outdoor testing to other countries because, according to a company source, “Sometimes we’re surprised and a product will have a unique sensitivity to the local environment that we just didn’t predict.”
New polymers, variations on traditional polymers (such as metallocene grades), and new copolymer blends and alloys are marketed aggressively for their cost-performance or processing benefits. However, durability of these new materials remains a question mark without adequate testing. Similarly, new colorant systems based on new organic pigments and pigment blends are replacing heavy-metal based colorants. These new colorants can have unexpected color stability and lightfastness problems.
In one real-life case, a pigmented plastic furniture part showed severe fading and hue shift after short exposure to sunlight entering through window glass. The colorant system consisted of inorganic white and black pigments as well as organic red, blue, and yellow. The product first exhibited overall fading, and then the hue shifted to tan and then green as the organic colorants lost saturation at different rates.
Indoor uv exposure may not always be recognized as a significant degradation factor. That proved true in the case of a polypropylene fluorescent lamp fixture, which discolored and became chalky and brittle due to uv exposure coming from adjacent lamps.
Many new colorant systems are delivered in concentrate form with a polymer carrier. The addition of this carrier to other base polymers can affect the weathering performance of the system. Also, pigments stable in the carrier may not be as stable in the base polymer. This is especially true for both rigid and plasticized PVC, where many pigments are incompatible with the high acidity caused by PVC’s gradual dehydrochlorination outdoors.
“Same-spec” resin from different plants, even different reactors, of the same supplier can differ in crosslinking, molecular-weight distribution, side-chain branching, or pellet size. All of these can affect processing and performance—including weatherability. This is especially true for semicrystalline polymers such as polypropylene. In one case the only apparent distinction between batches of good and bad product was a measurable increase in calcium content in the resin. Upon investigation, it was determined that this occurred when the resin supplier began shipping from a different plant, and the calcium difference was attributed to the local water supply used in the pelletizing operation. The change affected the weathering properties of the resin.
Fillers and other bulk additives are frequently bought on price, but when non-technical buyers switch vendors or grades to save a penny, they can compromise product performance. Talc, for example, can absorb HALS uv stabilizers and reduce their effectiveness. And metal content in various silica fillers can catalyze degradation of PP unless it is adequately stabilized.
Stabilizers or ‘unstabilizers’?
In a sense, all polymer additives can be thought of as contaminants. Despite their positive value, they can also have undesirable side effects. For example, titanium dioxide is used to protect resins like rigid vinyl from uv. However, TiO2 is also well known to cause polymer degradation in the presence of uv and moisture, resulting in chalking in the case of vinyl siding and window and door profiles. Fortunately, chalking can be avoided by using a weatherable grade of TiO2 with an appropriate surface coating.
Carbon black is another pigment often added for uv protection. However, carbon black comes in many grades and forms. Some of them have higher levels of surface functional groups that can adsorb antioxidants and thereby decrease instead of increase overall stability.
Metal chelates such as calcium and zinc stearates are often added as processing stabilizers. However, they can have antagonistic effects on other additives, such as hindered amine light stabilizers (HALS). The result can be diminished weatherability.
Hindered phenolic antioxidants are added to many polymers as processing stabilizers. However, they may react with atmospheric nitrogen oxides during the product’s service life to produce yellowing or pinking. Secondary stabilizers are needed to minimize this “gas fade” problem.
This effect is accentuated when the material is exposed to excessively high temperatures. For example, a manufacturer of picnic ice chests discovered it had a warehouse full of coolers with splotchy pink discoloration of the white PVC inner liner. The problem was that the warehouse reached temperatures of 100 F in summer. Similar color problems have frequently been encountered from overly hot processing of automotive TPOs.
Recycling adds risks
Addition of reground process scrap or post-consumer recyclables can severely affect product durability, particularly physical performance. These materials’ previous heat histories can deplete levels of protective antioxidant additives. The end result is then higher uv photosensitivity and decreased molecular weight, often accompanied by yellowing. Mechanical properties can also be compromised. To cite one example, a manufacturer of HDPE plastic deck lumber discovered that as antioxidants become depleted from reprocessing, the product’s weatherability performance declined rapidly.
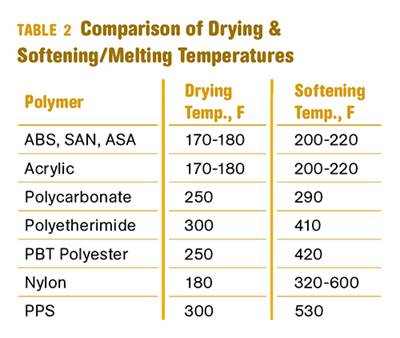
The risks associated with adding already process-degraded material to virgin resin are greater when it is not done on a consistent basis. Some processors start adding regrind only when the scrap pile builds up too high. The next parts out of the machine can contain nearly 100% regrind. Moreover, processors don’t always take the same care in drying regrind as they do the virgin resin. The added moisture content can have significant effects on polymers such as nylon, PET, and polycarbonate. This moisture, together with heat from processing and oxygen from the air, can form hydroperoxides, which can accelerate thermal degradation and sensitize the plastic to light exposure.
Government regulations, such as the European Community’s new “end-of-life” recycling directive for automotive materials, will soon start to have an impact on U.S. manufacturers. As the amount of recycled content increases, so will the potential consequences for long-term durability.
Tests validate processing
Still another reason to submit products to weathering tests is to ensure that improper processing does not compromise in-service durability. Premature yellowing of vinyl siding processed at overly high temperatures is just one example. Many product failures are traced to practices such as increasing the molding temperature to process a “difficult” batch of resin or increasing the extruder speed and die-head temperature to boost output. Such steps negatively affect the polymer by initiating free-radical oxidation and auto-catalytic degradation mechanisms, depleting antioxidants and sensitizing the polymer to uv attack.
Testing can also validate secondary processing procedures. For example, polycarbonate auto headlamp covers require hardcoats to protect against both scratches and uv-induced yellowing. But an improperly applied hardcoat can delaminate after exposure to the weather.
Possible deleterious effects of secondary processing may be overlooked in formulating a product for in-service durability. In one case, a manufacturer of injection molded PVC garage doors was using a proven compound and had tight control over the molding operation. However, the door segments had mitered corners that were ultrasonically butt welded. Though the PVC was stabilized for initial processing and normal weathering, the welding process caused thermal degradation at the butt joints that resulted in severe yellowing and cracking upon outdoor exposure.
Many plastics are coated for performance or appearance properties. These coatings can sometimes have negative effects on plastics, such as increased yellowing. New golf balls for example, often have an anti-slip coating to minimize air drag, but the coating may cause discoloration of the plastic substrate.
Risk avoidance
It is neither feasible to design products to be fail-safe nor to test for all possibilities. However, there are usually a limited number of identifiable variables that affect product durability. Therefore, it is prudent for material producers and processors to conduct basic designed experiments to gauge the sensitivity of the product to formulation, processing, and environmental factors. Statistically designed experiments are far more productive and cost-effective than the “test it and see what happens” approach.
Even a modest durability testing program can usually pay for itself by decreasing warranty costs and allowing processors to optimize formulations and processing methods without compromising product weatherability.
A change in resin, additive, or processing can lead to unpleasant surprises in weathering performance if testing is not performed. (Shown here: outdoor testing in Florida)
Strips cut from injection molded PP roofing shingles show the effect of substituting an inferior carbon black that adsorbed the antioxidants and left the parts under-stabilized. Section on right is unweathered. Its mottled appearance is meant to imitate natural slate. Part on left shows premature surface degradation.
Allen F. Zielnik is director of technical strategic sales and durability technical consulting at Atlas Material Testing Technology LLC in Chicago. Atlas supplies Weather-Ometer and Xenotest weather-testing instruments as well as outdoor weather testing services through its Atlas Weathering Services Group. Atlas can be reached at (773) 327-4520, www.atlas-mts.com.
Related Content
Robust Rotational Viscometer
Versatile viscometer line said to offer robust measurement, repeatability and reporting.
Read MoreX-Ray Vision Inside Parts Gets More Affordable for Processors
Shimadzu’s new benchtop x-ray CT scanner provides internal and external metrology and flaw detection at a fraction of the previous cost.
Read MoreDatacolor Acquires Matchmycolor
The acquisition of the specialist in color formulation and communication software further expands Datacolor’s global industry presence in color management.
Read MoreFast, Simple QC Method Directly Quantifies Recycled Content in Plastics
Novel fluorescence-based spin-out technology provides a rapid and reliable method to measure and certify recycled content in a wide range of plastics.
Read MoreRead Next
How Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read MoreWhy (and What) You Need to Dry
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreLead the Conversation, Change the Conversation
Coverage of single-use plastics can be both misleading and demoralizing. Here are 10 tips for changing the perception of the plastics industry at your company and in your community.
Read More