Dimensional Stability After Molding—Part 2
After molding, acetal parts can continue to shrink at room temperature and even in the cold.
My first exposure to the prolonged shrinkage process exhibited by some materials came when I was molding large parts in acetal homopolymer. Most of my experience with molding acetal parts to close tolerances had come from producing small gears with diameters of no more than half an inch.
However, our company landed a project that involved housings and quadrant gears with critical dimensions in the range of 3.5 to 4 in. and print tolerances of ±0.010 in. on parts with nominal wall thicknesses of 0.110 in. One specific dimension governing the spacing of alignment holes called for a specification of 4.046 ±0.010 in.
During initial sampling we produced parts with measurements that ranged from 4.038 to 4.042-in. in a 30-piece capability study. Statistical process control (SPC) was a relatively new concept in U.S. manufacturing at that point, so the fact that we were operating at one end of the tolerance range did not particularly concern us. We were quite satisfied that the dimensional range was tight and all the parts were to print.
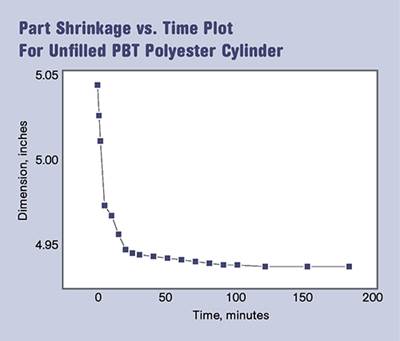
But the parts had been measured about 90 minutes to two hours after they were produced. The parts were room temperature to the touch and based on our experience with other semi-crystalline materials we were satisfied that everything was fine. The next day the quality assurance people pulled us into the lab to show us that half of the parts we had produced the day before were too small to meet the print. A review of all 30 parts showed that they had all continued to shrink and were now 0.004 in. smaller than the previous afternoon. In another day they moved an additional 0.001 in. and then things seemed to settle down. Later I observed similar problems with large parts produced in polypropylene, even in filled PP.
In order to understand what was happening it is important to appreciate the relationship between mold shrinkage and crystallization in semi-crystalline materials. The more the material crystallizes the more it shrinks. Optimal levels of crystallinity are desirable. Semi-crystalline materials offer improved levels of fatigue and wear resistance compared with amorphous polymers and they generally provide improved creep resistance at elevated temperatures. But if the material is molded in a manner that prevents the development of crystallinity, these properties are not realized at the intended levels.
The opportunity for crystallization exists in a temperature window below the melting point of the polymer and above the glass-transition temperature (Tg). Processes like injection molding involve rapid cooling of the polymer as it enters the mold. Even when running a material such as PEEK, where the mold temperature may be 375 F (190 C), this represents a thermal shock to the flowing polymer that enters the mold at 700 F (371 C). This rapid reduction in temperature is needed to solidify the material so that the part can assume its intended shape. But as long as the polymer remains at a temperature above its glass transition, about 295 F (146 C) for PEEK, there will be sufficient mobility at a molecular level to allow the crystal structure to develop. Once the temperature of the material drops below this point no more crystals can form.
So what happens with acetal and PP? I often hear people state that these materials do not follow the rules. The rule they are referring to is that if the part feels like it is at room temperature then it is dimensionally stable. In fact, these materials follow the rules precisely. The problem is that the Tg of these materials is below room temperature. This is also true of polyethylene and ethylene copolymers such as EVA. The Tg of PP can vary from about -10 C to +15 C (14 to 59 F) depending upon the grade. The Tg of acetals is -78 C (-109 F), or the temperature of dry ice. This is a temperature only rarely experienced anywhere on our planet. So the chances are quite good that wherever you might be using an acetal part, it is above its Tg. Room temperature is 100° C above the Tg of acetal. So as our molded parts sat in the quality lab overnight, they had more than enough freedom at a molecular level to continue crystallizing.
A properly packed out acetal part produced at the correct mold temperature will exhibit continued shrinkage of about 0.001 in./in. between the time the part reaches room temperature and the time that it is truly stable. This value may increase if the nominal wall is very thick. For small parts this change will be difficult to detect unless very precise measurements are made. But in larger parts it becomes a significant factor. Even after the dimensions stop changing at a measurable level, structural rearrangements continue to occur for weeks. Several years ago we studied the tensile strength and modulus, impact resistance, and dynamic mechanical properties of molded PP and acetal specimens over a period of five weeks after molding.
Properties were measured 48 hr after parts were produced, which is the ASTM guideline, and then additional tests were performed at intervals of seven days out to 35 days after the molding date. Tensile strength and modulus continued to rise over time, although the rate of increase declined as the time extended. Impact performance became worse. Some materials that were ductile two days after molding began to exhibit brittle behavior in 7 to 14 days. All of this was the direct result of continued crystallization.
Most processors do not consider the effect of processing conditions such as mold temperature on part performance. They are much more concerned with making parts that are to print. So when a process produces parts that are undersized or on the low end of the tolerance range, processors think in terms of making adjustments to correct this condition. The adjustment of choice is almost always mold temperature. Reduce the mold temperature and the part will be larger. The reason is simple: A lower mold temperature limits the ability of the polymer to crystallize. Fewer crystals mean less shrinkage.
But in a material that has a Tg below room temperature, these gains may be temporary. In every material there is an ideal spacing between molecules. The material “knows” what this spacing is and given the opportunity it will do everything it can to achieve it. Suppressing crystallinity in a material that possesses structural mobility at room temperature is a short-term strategy. This is why the suppliers of acetal materials have been so insistent over the years on the importance of mold temperature and proper packing to prevent something they call post-mold shrinkage.
It turns out that dimensional changes can also occur due to another mechanism that primarily affects amorphous materials, a class of polymers that is considered to have the upper hand when it comes to dimensional stability. However, these changes take much longer to unfold and they are smaller in magnitude. But if you are really focused on very close tolerances and the parts are large enough, these changes may still be of concern. We will deal with this mechanism in our next installment.
Editor’s note: You can read the next part in the series by clicking here.
Related Content
Volume Resin Prices Move in Different Directions
PE, PP, PVC, and ABS prices slump, while PS, PET, PC, and nylons 6 and 66 prices rise.
Read MoreTracing the History of Polymeric Materials: Polyphenylene Oxide
Behind the scenes of the discovery of PPO.
Read MoreTracing the History of Polymeric Materials: Acetal
The road from discovery in the lab to commercial viability can be long, and this was certainly the case for acetal polymers.
Read MoreHow Do You Like Your Acetal: Homopolymer or Copolymer?
Acetal materials have been a commercial option for more than 50 years.
Read MoreRead Next
Dimensional Stability After Molding: Part 1
The degree to which molded parts shrink as they cool is largely dependent upon the composition of the material being processed.
Read MorePeople 4.0 – How to Get Buy-In from Your Staff for Industry 4.0 Systems
Implementing a production monitoring system as the foundation of a ‘smart factory’ is about integrating people with new technology as much as it is about integrating machines and computers. Here are tips from a company that has gone through the process.
Read MoreTroubleshooting Screw and Barrel Wear in Extrusion
Extruder screws and barrels will wear over time. If you are seeing a reduction in specific rate and higher discharge temperatures, wear is the likely culprit.
Read More
.jpg;width=70;height=70;mode=crop)











.png;maxWidth=300;quality=90)














.png;maxWidth=970;quality=90)