MATERIALS: The Importance of Oxidative Stability in Polyolefins
Because oxidation is a process that causes materials to deteriorate over time, its effects or potential for those effects are not always apparent when new products are tested.
Polyethylene and polypropylene are converted into billions of pounds of products every year by essentially every process used in plastics. They are low-cost materials that are also among the lightest materials in existence, therefore the cost per unit weight (lb or kg) gets even better when the calculation is made as cost per unit volume (in.³ or cm³). They process easily, are not affected by moisture, and have good electrical properties and good-to-excellent chemical resistance.
But like all materials they have their weaknesses, and one of these is susceptibility to oxidation. Polypropylene (PP) is more vulnerable than polyethylene (PE) due to some important differences in chemistry, but both materials rely on the incorporation of additives known as antioxidants in order to survive the elevated temperatures of melt processing and the demands of various application environments.
Because oxidation is a process that causes materials to deteriorate over time, its effects or potential for those effects are not always apparent when new products are tested. Most PPs and PEs are quite ductile as molded, so brittle failure is not typically considered to be a likely outcome. But anyone who has conducted oven-aging studies on PP has witnessed how quickly a tough, flexible material can turn brittle. While the elevated temperatures of these tests may not compromise the short-term properties in an obvious manner, they do increase the rate at which the antioxidants are consumed. Once they are gone, the rate of decline in performance can be dramatic.
All PPs melt between 150 C and 165 C (302-329 F), depending upon whether they are homopolymers or copolymers. Most of the attention regarding elevated-temperature performance focuses on retention of strength and stiffness as temperature increases. But exposure to elevated temperatures has a long-term effect that can ambush an end user because the level and type of antioxidant that is incorporated into the material may limit the ability of the material to function at even mild temperatures over the long term.
The UL rating known as relative temperature index (RTI) is intended to capture this property of oxidative stability. Higher RTI ratings indicate that a material is more resistant to oxidation and can survive for longer periods of time at elevated temperatures. This is all about the stabilizers in the material, which can constitute less than 0.1% of the compound by weight. An examination of UL “yellow cards” for various PPs will show that some materials in this family have RTI ratings as high as 125 C (257 F), relatively close to the melting point of the material, while others only register a value of 60-70 C (140-158 F). The big reason for this difference is the level of protection afforded to the polymer by the antioxidant package.
This antioxidant package is typically a mixture of different constituents that work together to protect the material from the effects of elevated temperatures. Melt processing involves the most extreme conditions, with temperatures ranging from as low as perhaps 135 C (275 F) for a thick-walled LDPE part to 288 C (550 F) or higher for some high-speed film-processing lines. But this exposure is expected to be brief—a matter of a few minutes. Some antioxidant will be consumed during processing, so the ability of the final product to function in the application environment is a matter of how much protection the raw material started with and how well it was preserved by the processor. Elevated temperatures and long dwell times take their toll in ways other than immediate thermal degradation.
Once the part has been fabricated, then the environment takes over. The temperature of the application and the duration of that exposure to temperature are the most obvious concerns. A part such as a bottle that holds a consumable product and serves simply as packaging may never experience anything more challenging than the elevated temperatures of a shipping container followed by a few weeks in a warehouse, then on a store shelf, and finally in someone’s kitchen or bathroom. But a part like a battery jar or a surge tank that holds antifreeze under the hood of an automobile may see temperatures over 100 C (212 F) for several thousand hours. The suitable level of antioxidant protection needed for these two applications will be very different.
Sometimes the application is demanding without appearing to be. A simple four-cup coffee maker contains multiple PP parts. Every time coffee is brewed, some of these parts will be exposed to not only heat but flowing water. Water is an excellent solvent, and hot water is a better solvent than cold water. Some antioxidants can be extracted by the heated water, consequently reducing the life of the components. And while the exposure time per use may be relatively short, use may occur daily for many years.
Many years ago, I worked on a problem that an end user was having with the spigots at the bottom of hotel and convention-center coffee makers that were breaking off when someone pulled the lever to dispense coffee. These spouts are typically made of PP, and while the as-molded part was quite tough, the failures were obviously very brittle. Laboratory analysis showed that the antioxidant in the material had been completely consumed; there was no more protection from the rigors of the application environment. The problem was traced back to a change that had been made to the specified raw material. The original material was formulated with an additive package designed to handle long-term exposure to the hot, wet conditions of the appliance. The new material, which of course was less expensive, contained significantly less antioxidant and reduced the expected life of the part by as much as 90%.
The failure-analysis literature is full of case studies involving PP and, to a lesser extent, PE components that turn brittle over time while exposed to elevated temperatures, with or without water contact. PP wire insulation that had been buried underground with the expectation that it would last 30-40 years failed in a much shorter time due to oxidation. This occurred many years ago when PP was still a fairly new material. When the cables were unearthed, it was found that the insulation was very brittle. The analysis indicated that the problem was caused by multiple influences that included ground water, the mechanical pressure of the soil and rocks, and the fact that copper catalyzes oxidation in polyolefins.
The scientists who worked on that problem discovered that they could assess the relative oxidative stability of the material in the laboratory. Using an instrument known as a differential scanning calorimeter (DSC), it was possible to place a sample in a controlled environment involving elevated temperature and an oxygen-containing atmosphere and observe the time required before the sample oxidized. Oxidation is a highly exothermic process in polyolefins, and the heat release associated with the process was readily detected.
In the next installment on this topic we will discuss these test methods, their uses and limitations, and how they can be used to diagnose problems and trace them back to their source.
ABOUT THE AUTHOR
Mike Sepe is an independent, global materials and processing consultant whose company, Michael P. Sepe, LLC, is based in Sedona, Ariz. He has 40 years of experience in the plastics industry and assists clients with material selection, designing for manufacturability, process optimization, troubleshooting, and failure analysis. Contact: (928) 203-0408 mike@thematerialanalyst.com.
Related Content
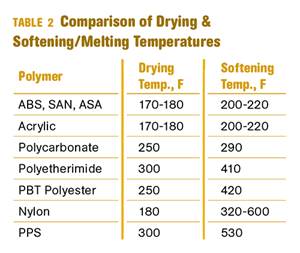
Why (and What) You Need to Dry
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreUnderstanding Strain-Rate Sensitivity In Polymers
Material behavior is fundamentally determined by the equivalence of time and temperature. But that principle tends to be lost on processors and designers. Here’s some guidance.
Read MoreA Simpler Way to Calculate Shot Size vs. Barrel Capacity
Let’s take another look at this seemingly dull but oh-so-crucial topic.
Read MoreThe Importance of Melt & Mold Temperature
Molders should realize how significantly process conditions can influence the final properties of the part.
Read MoreRead Next
Processor Turns to AI to Help Keep Machines Humming
At captive processor McConkey, a new generation of artificial intelligence models, highlighted by ChatGPT, is helping it wade through the shortage of skilled labor and keep its production lines churning out good parts.
Read MoreTroubleshooting Screw and Barrel Wear in Extrusion
Extruder screws and barrels will wear over time. If you are seeing a reduction in specific rate and higher discharge temperatures, wear is the likely culprit.
Read MoreUnderstanding Melting in Single-Screw Extruders
You can better visualize the melting process by “flipping” the observation point so that the barrel appears to be turning clockwise around a stationary screw.
Read More
.jpg;width=70;height=70;mode=crop)











.png;maxWidth=300;quality=90)