Chasing Nanocomposites
Nano-sized particles have mega-potential in plastics because just a pinch does so much more than heavy loadings of other additives. Three recent conferences presented almost 200 papers on the feverish pace of ‘nano’ R&D on boosting plastics’ mechanical and barrier properties, flame retardancy, and electrical conductivity.
They are still in their infancy, but if the forecasts are right, nanocomposites could turn out to be the biggest little thing to hit plastics in decades. Polymers reinforced with as little as 2% to 5% of these particles via melt compounding or in-situ polymerization exhibit dramatic improvements in thermo-mechanical properties, barrier properties, and flame retardancy. They also can outperform standard fillers and fibers in raising heat resistance, dimensional stability, and electrical conductivity.
Dispersions of nano-scale reinforcements in polymers are already entering the marketplace in automotive and packaging applications, albeit in a low-profile manner and slower than had been anticipated. But that pace is expected to speed up dramatically, as indicated by the enthusiasm of researchers and marketers shown in roughly 200 papers delivered at three technical conferences. These were Nanocomposites 2004 in San Francisco, the SPE Antec 2004 in Chicago, and Nanocomposites 2004 in Brussels, Belgium (see box for details).
A report from market-research firm Business Communications Co., Inc., Norwalk, Conn., pegs the total worldwide market for polymer nanocomposites at 24.5 million lb in 2003, valued at $90.8 million. It also projects the market to grow at an average annual rate of 18.4% to reach $211.1 million by 2008. Even if nano developments hit some snags, BCC says some applications will grow faster than 20% per year.
The leading nano-scale fillers in R&D and commercial projects are layered silicate nanoclays and nano-talcs, plus carbon nanotubes and graphite platelets. But other candidates are being actively investigated, such as synthetic clays, polyhedral oligomeric silsesquioxane (POSS), and even natural fibers like flax and hemp.
The top contenders
The two types of nano-fillers that have been most widely discussed and the first to break into commercial use are nanoclays and carbon nanotubes. Both must be chemically modified with surface treatments in order to achieve the fine dispersion and resin coupling that are required to derive maximum benefit.
Both of these nano-fillers have demonstrated improvements in structural, thermal, barrier, and flame-retardant properties of plastics. Carbon nanotubes also enhance electrical conductivity.
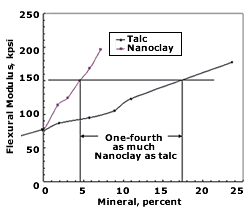
So far, nanoclays have shown the broadest commercial viability due to their lower cost—$2.25 to $3.25/lb—and their utility in common thermoplastics like PP, TPO, PET, PE, PS, and nylon.
The leading nanoclay is montmorillonite, a layered alumino-silicate whose individual platelets measure around 1 micron diam., giving them an aspect ratio of 1000:1. The two major domestic producers are Nanocor with its Nanomer line and Southern Clay Products with its Cloisite line. Both companies have formed alliances with suppliers of resins and surfactants, plus compounders and automotive OEMs and packaging firms. While much of their work is proprietary, they have disclosed several commercial successes.
General Motors has taken the lead in putting nanocomposites on the road. GM launched the first commercial auto exterior use of a nanocomposite in the step assist on the 2002 GMC Safari and Chevrolet Astro van. The part also appears on 2003 and 2004 models. More recently, a PP/nanoclay composite appeared on the body side molding of General Motors’ highest-volume car, the 2004 Chevrolet Impala. The compound was developed by GM’s R&D Center in Warren, Mich., in cooperation with Basell North America and Southern Clay Products.
The latest application is on the 2005 GM Hummer H2 SUT. The vehicle’s cargo bed uses about seven pounds of molded-in-color nanocomposite parts for its center bridge, sail panel, and box-rail protector. The material is Basell’s Profax CX-284 reactor TPO with nanoclay.
While nanoclay adds muscle to plastics, carbon nanotubes impart electrical and thermal conductivity. Nanotubes’ commercial potential has been limited by their high price tags—reportedly in the range of $100/gram, although they are available in masterbatches for $50/lb and up. Still, nearly every car produced in the U.S. since the late 1990s contains some carbon nanotubes, typically blended into nylon to protect against static electricity in the fuel system. Static-dissipative compounds containing nanotubes are also protecting computer read/write heads.
Carbon nanotubes include both single-and multi-walled structures. The former have a typical outside diameter of 1 to 2 nm while the latter have an OD of 8 to 12 nm. They can range in length from the typical 10 microns to as much as 100 microns and have at least a 1000:1 aspect ratio. Carbon nano tubes have 50 times the tensile strength of stainless steel (100 GPa vs. 2 GPa) and five times the thermal conductivity of copper. When incorporated into a polymer matrix, they have the potential to boost electrical or thermal conductivity by orders of magnitude over the performance possible with traditional fillers such as carbon black or metal powders.
Domestic suppliers of nanotubes include Hyperion Catalysis with its Fibril multi-walled nanotubes and newcomer Zyvex Corp. with its NanoSolve single- or multi-walled tubes. Both suppliers now offer their products in masterbatches that typically contain 15% to 20% nanotubes.
A different but related category is vapor-grown carbon nano-fibers from Pyrograf Products, a spin-off from Applied Sciences. Its Pyrograf III nano-fibers reportedly can compete with nano-tubes in providing thermal and electrical conductivity and dramatically enhancing mechanical properties and fire resistance (char formation). What’s more, nano-fibers cost significantly less—around $100 to $150/lb. Evaluations are under way in nylon, PP, and polyurethanes.
Nanoclay compound sources
Alliances between Nanocor and two specialty compounders, have resulted in commercial-scale nanocomposite concentrates and compounds for structural and barrier applications.
Noble Polymers new Forte PP nanocomposite compound made its commercial debut in the seat backs of the Honda Acura TL 2004 car. Forte replaced glass-filled PP, which caused processing difficulties, visual defects, and warping. Forte has a low density of 0.928 g/cc, superior mechanical properties, and improved surface quality and recyclability.
Noble reports that the Forte nanocomposite will also be used to produce the center console for a 2006 model light truck. Other applications in the works include office furniture—replacing 20% glass-filled PP—and appliance parts, where Forte reduces weight and possibly material cost.
PolyOne recently introduced the Maxxam LST line of PP homopolymer/nanoclay compounds that boast high stiffness and impact resistance. Through a patent-pending process, PolyOne reports that it has been able to overcome previous problems of incomplete exfoliation and dispersion of the nanoclay, resulting in performance that meets or exceeds many engineering thermoplastics. Lighter weight, aesthetic and processing advantages, and lower cost are also claimed.
PolyOne also offers Nanoblend concentrates of up to 40% nanoclay in homopolymer PP, modified PP, LLDPE, LDPE, HDPE, or an ethylene copolymer. Some grades are tailored specifically for barrier enhancement.
PolyOne reports that applications nearing commercialization include pallets and dunnage, where Maxxam LST compounds are specified as alternatives to engineering resins due to their improved dimensional control, which is critical for robotic assembly. In addition, they boast good impact strength and lighter weight. Maxxam LST is also being considered for consumer disposable applications due to a combination of chemical resistance and stiffness, as well as dramatic cycle-time improvements.
Meanwhile, the Nanoblend concentrates are being considered for auto interior and exterior TPO parts. Key drivers are dimensional stability, lighter weight, and stiffness without loss of impact. Nanoblend concentrates are being evaluated in films for enhancing barrier, stiffness, HDT, and controlled release or migration of additives such as biocides and dyes. In blow molded packaging, Nanoblend is being considered for improved barrier and the potential for thinwalling and faster cycles. Thin-walling and faster cycles are also attractions in injection molded containers and totes. Some industry sectors are evaluating the concentrates for improving flame retardancy.
More muscular TPOs
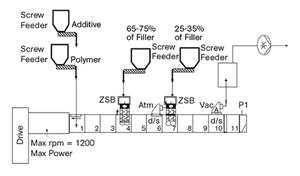
Papers presented by General Motors and Southern Clay Products discussed numerous enhancements to automotive TPOs obtained with nanoclays. Those advances did not come easily: Early processing problems caused by clay agglomeration were ultimately resolved by optimizing the clay infeed position at the extruder, the screw design, screw speed, temperature, and pressure. Once processing issues were re solved, nanocomposite TPOs outperformed conventional talc-filled TPOs in consistency of properties, retention of low-temperature ductility, elimination of “tiger striping,” reduced paint delamination, and improved knit-line appearance, colorability, grain patterns, scratch and mar resistance, and recyclability. What’s more, the lower filler level means 3% to 21% lower density (0.92 vs. 0.96 to 1.13 g/cc). Lighter weight requires less adhesive for attachment, which cuts cost.
Among the many auto exterior, interior, and under-hood applications for which nanocomposites appear suited are fascias, rocker covers, side trim, grilles, hood louvers, instrument panels, seat/IP foams, door inners, pillar covers, vertical and horizontal body and closure panels, engine shrouds, fan shrouds, air intakes, fuel tanks, and fuel lines.
In addition to TPO/nanoclay composites, GM has explored using carbon nanotubes to replace current thermoset structural composites. GM is interested in reducing reinforcement levels in Class A applications by replacing continuous carbon fibers with nanotubes or short nano-fibers. Nanotubes also have the potential to reduce plastics’ coefficient of thermal expansion more effectively.
Better barriers
Polymer barrier technology is also getting a boost from nanoclays. Mitsubishi Gas Chemical (MGC) and Honeywell Specialty Polymers both are using Nanocor’s nanoclays in nylons as barrier layers in multi-layer PET bottles and films for food packaging. MGC’s MXD6 nylon nanocomposite, called Imperm N, is used commercially in Europe in multi-layer PET bottles for beer and other alcoholic beverages. It is also being evaluated for small carbonated soft-drink bottles. Other Imperm applications that will debut in the next six months are multi-layer thermoformed containers for deli meats and cheeses and flexible multi-layer films for potato chips and ketchup.
Honeywell has aimed its Aegis nylon 6 nanocomposites initially at PET beer bottles. In late 2003, a version containing an oxygen scavenger made a commercial splash with the introduction of the 1.6-liter Hite Pitcher beer bottle from Hite Brewery Co. in South Korea. Aegis is the barrier layer in this three-layer structure, which is said to provide a 26-week shelf life.
Honeywell is aiming other Aegis nanocomposite grades (without oxygen scavenger) as replacements for EVOH in films and pouches. Such grades reportedly are lower in cost than EVOH, provide a better barrier that allows for lightweighting, and also have better puncture resistance and good clarity. (Because of their size, nano-particles do not interfere with light transmission.)
The U.S. military and NASA, in conjunction with Triton Systems, Inc., Chelmsford, Mass., are looking into nanoclay as a barrier enhancer for EVOH in long-shelf-life packaging. An experimental thermoformed food tray was made from EVOH plus 3% of Southern Clay’s Cloisite in a layer sandwiched between two PP layers. It reportedly imparts three- to five-year shelf life without refrigeration, plus good clarity, processability, and recyclability.
Alcoa CSI, Crawfordsville, Ind., is seeking a patent on coextruded barrier liners for plastic bottle caps for beer, juice, or carbonated soft drinks. The liners include a layer of nylon 6/nano clay composite plus one or two EVA layers with oxygen scavengers. This liner is said to outperform other barrier materials at very high humidity (95% to 96% RH).
LG Chem Ltd. of South Korea has developed high-barrier, monolayer blow molded containers of HDPE with 3% to 5% nanoclay for handling toluene and light hydrocarbon fluids. LG reports that permeation of the hydrocarbon solvents is cut by a factor of 40 to 200 compared with neat HDPE.
Versatile nanocarbons
Since the early 1990s, automotive fuel-line components such as quick connectors and filters have used inner barrier layers consisting of nylon 12 and carbon nanotubes. Hyperion Catalysis now aims to introduce nanotubes into other resins used in auto fuel systems, such modified nylons and fluoropolymers. A new fluoropolymer/nanotube compound is being used to make O-rings for automotive fuel connectors.
In electronics, polycarbonate and polyetherimide (GE’s Ultem) components of computer hard drives have been reinforced with nanotubes to render them conductive and very smooth.
Over the last three years, a major automotive OEM in Europe has been using carbon nanotubes in GE’s Noryl GTX nylon/PPO alloy to mold exterior fenders. This conductive nanocomposite allows for electrostatic painting.
Michigan State University’s Composite Materials and Structures Center in East Lansing developed a new surface-treated graphite nano-platelet. Graphite has a modulus several times that of clay and also has excellent electrical and thermal properties. When incorporated into an epoxy, it results in superior mechanical properties and excellent electrical conductivity compared with standard carbon fibers and nano-sized carbon black. MSU sees potential in ESD protection and EMI shielding. Plastic nano-graphite compounds are expected to sell for up to $5/lb, significantly less than compounds based on nanotubes or vapor-grown carbon fibers.
Carbon nanotubes have more going for them than just conductivity. Researchers at the National Institute of Standards and Technology (NIST), Gaithersburg, Md., report that carbon nanotubes in PP not only enhance the material’s strength and properties, but also dramatically change how the molten polymer flows, virtually eliminating die swell.
Nano flame retardants
Extensive research at NIST has established nanoclays’ effectiveness as flame-retardant synergists. Nanoclay levels of 2% and 5% in nylon 6 reduced the rate of heat release by 32% and 63%, respectively, NIST found.
Specialty compounder Foster Corp. recently demonstrated that higher levels (13.9%) of nanoclay can be added to nylon 12 elastomers to achieve UL 94V-0 rating at 1/8-in. thickness. Used as a char former, the nanoclay allows the typical 50% loading of halogen/antimony oxide flame-retardant system to be cut in half, which significantly reduces detrimental effects on physical properties. The company first introduced nylon 12/nanoclay compounds for tubing and film in 2001.
Germany’s Sud-Chemie (U.S. office in Louisville, Ky.) offers modified nanoclays called Nanofil as flame retardants. It recently developed halogen-free EVA/PE wire and cable compounds containing 3% to 5% of new Nanofil SE 3000 plus 52% to 55% alumina trihydrate or magnesium hydroxide (typically used at 65% levels). The result is said to be improved mechanical properties, smoother cable, and higher extrusion speeds.
According to Hyperion Catalysis, two recent studies show that multi-walled carbon nanotubes may act as a flame retardant without use of halogen. In both EVA and maleic-anhydride-modified PP, 2.4% to 4.8% loadings of nanotubes show heat-release rates comparable to or better than those obtained with nanoclays.
Nano-nucleators
Among its many virtues, nanoclay can work as a nucleating agent to control foam cell structure and enhance properties of polymeric foams for applications from insulation to packaging. The University of Toronto’s Dept. of Mechanical and Industrial Engineering studied extrusion of chemically foamed LDPE/wood-fiber compounds. Addition of 5% nanoclay to the mix decreased the cell size, increased the cell density and facilitated foam expansion. When burned, the foam showed good char formation. Similar results were obtained in LDPE/nanoclay foam blown with CO2 gas.
Researchers at Ohio State University’s Dept. of Chemical Engineering (Columbus) found that small amounts of nanoclay surface-grafted with PMMA can reduce cell size and increase cell density in microcellular PS foamed with CO2. Another OSU study showed that smaller cell size and higher density can be achieved with 5% nanoclay in polyurethane foams blown with pentane or water.
Louisiana State University’s Mechanical Engineering Dept. (Baton Rouge) reports that 4% to 5% nano clay increases the flexural strength and elongation of epoxy syntactic foams used as core materials for sandwich composites in structural applications.
Related Content
Green’s the Theme in Extrusion/Compounding
The drive toward circular economy is requiring processors to make more use of PCR. Machine builders at K—across all extrusion processes—will be highlighting innovations to help them do just that.
Read MoreImproving Twin-Screw Compounding of Reinforced Polyolefins
Compounders face a number of processing challenges when incorporating a high loading of low-bulk-density mineral filler into polyolefins. Here are some possible solutions.
Read MoreHow to Maintain Pelletizing Quality When Acid Attacks
Developments in the chemistry of polymers and additives have made corrosion a real problem in pelletizers. Here’s how to ward it off.
Read MoreLFT-D Thrives in Automotive and Other Durables
Teijin Automotive acquires its 10th direct long-fiber thermoplastic system as demand for this technology soars.
Read MoreRead Next
Understanding Melting in Single-Screw Extruders
You can better visualize the melting process by “flipping” the observation point so that the barrel appears to be turning clockwise around a stationary screw.
Read MoreLead the Conversation, Change the Conversation
Coverage of single-use plastics can be both misleading and demoralizing. Here are 10 tips for changing the perception of the plastics industry at your company and in your community.
Read MoreProcessor Turns to AI to Help Keep Machines Humming
At captive processor McConkey, a new generation of artificial intelligence models, highlighted by ChatGPT, is helping it wade through the shortage of skilled labor and keep its production lines churning out good parts.
Read More.png;maxWidth=970;quality=90)





















.png;maxWidth=300;quality=90)