Getting Started in LSR: Understanding the Materials, Part I
Liquid silicone rubber (LSR) injection molding is a long established process but it is enjoying an upsurge in interest for medical, automotive, infant care, and general industrial applications.
Liquid silicone rubber (LSR) injection molding is a long established process but it is enjoying an upsurge in interest for medical, automotive, infant care, and general industrial applications. LSR is a thermoset that offers properties not obtainable with today’s TPEs. LSR has attracted growing attention for its heat resistance, extreme low-temperature flexibility, chemical resistance, biological inertness, and inherent lubricity. And owing to the growing popularity of hard/soft overmolding, many thermoplastic molders are looking at LSR molding as a way to expand the scope of their business. Luckily, there are many similarities between LSR processing and that of thermoplastics. With the proper training and attention to the differences that do exist, many thermoplastic molders have made a successful transition to LSR.
WHAT IS LSR?
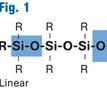
The basic raw material for silicone rubber is sand, or silicon dioxide. Sand is processed into pure silicon metal and then is reacted with methyl chloride, followed by an additional series of steps to create the many forms of silicone, from fluids and polymers to finished rubber bases and compounds. Unlike most plastics, silicones have a backbone of alternating atoms of silicon and carbon, with organic side chains that impart curability and other properties (see Fig. 1). One such form, LSR, has some processing characteristics in common with thermoplastics, along with some notable differences that are inherent in a thermoset.
LSR’s are crosslinked utilizing a platinum-catalyzed reaction that produces no by-products. Once cured, LSR cannot be reshaped or easily reused or recycled. To do so requires a great deal of energy to overcome and break apart the crosslinks and/or the backbone itself. This same molecular structure makes LSR useful over a wide range of temperatures. At the same time, the strong bonds between the silicon and oxygen atoms mean that the polymer does not degrade until heated to temperatures above those that most other polymers can withstand. Another of LSR’s major attributes is its chemical inertness or purity. Coupled with their ability to withstand sterilization processes, this makes LSR’s ideal for many medical uses as well as for baby care.
HOW DOES IT PROCESS?
With this in mind, we should look at some of the similarities and differences of how LSR is processed compared with thermoplastics. At first glance, an LSR injection machine may look quite like a thermoplastic injection press. Both have feeding systems that move the material into the injection barrel, which then moves the material into the mold in order to take the shape of the finished part. One of the largest differences occurs within the mold. While thermoplastics must be heated in the injection barrel to their melting point and then cooled in the mold back to a solid state, the reverse occurs with LSR. It starts out as a fluid and is then heated within the mold to initiate curing. The injection barrel is water-cooled to keep the temperature below the point where this reaction will progress. The mold is heated normally at 180 to 200 C, or 355 to 390 F. Whereas thermoplastic tooling can employ hot runners to minimize sprue and runner scrap by keeping them molten between shots, LSR employs cold-runner systems to keep the material cold until it enters the hot cavity. Because LSRs are high-value materials that cannot be reused, minimizing scrap is even more important than with thermoplastics.
The means of feeding LSR to the injection machine is another major difference between this material and thermoplastics. LSR comes as a paste with a consistency similar to that of peanut butter. It normally comes in two components—one containing the crosslinker and the other the catalyst—that are highly stable when separate but must be mixed together in order to allow the curing reaction to occur. To get to the injection barrel, the “A” and “B” components must be pumped into the injection barrel at a 1:1 ratio and they must be thoroughly blended prior to injection. This is usually accomplished with a water-cooled static mixer in a chamber prior to the injection unit. Most LSR injection machines also use an injection screw that contributes additional mixing.
Compared with thermoplastics, LSR materials in a hot mold tend to have very low viscosity, which requires careful attention by toolmakers and molders to parting lines and venting in order to prevent flash. On the plus side, this makes LSRs able to fill very long, thin sections easily.
Thermoplastics must be “packed” into the mold to ensure proper fill-out of the entire shape of the part. Packing pressure allows for additional material to be injected as the part shrinks. This is not an issue with LSR. One result of the crosslinking reaction is thermal expansion. Packing would result in excessive expansion when the mold opens, leading to uncontrollable part dimensions. Upon cooling, silicone rubber also contracts, but with a consistent shrinkage rate. For these reasons, silicone molding utilizes a hold pressure with molds designed to take both the expansion and the shrinkage rate into account for final desired dimensions.
Processing of LSR should be looked upon as a four-component system. The type and grade of LSR material is only the first of these. The quality of the injection machine, pumping unit, and mold also play vital roles. With proper planning and technique, it is possible to run an LSR part as fully automatic as a thermoplastic part.
MANY VARIETIES OF LSR
Like thermoplastics, there are many different types and grades of LSR. They can have a “wet” (rubbery) or “dry” (slippery) feel and a range of hardnesses. General-purpose grades are not highly filled (typically with silica) and are suitable for applications that need only basic physical properties. With the addition of additives and other fillers, LSR can be formulated to withstand higher temperatures or oil and other fluid environments. The addition of phenyl units stretches the low-temperature capabilities of LSR. Phenyl fluid can be added to reduce the coefficient of friction, making the surface of the part slippery as the fluid bleeds out over time. There are also versions available that impart low friction chemically, without the need for fluid bleeding to the surface.
Of great importance to many plastic molders and designers are the newer grades of self-adhesive LSR for hard/soft overmolding applications, also called two-component or 2K molding. These materials no longer require a separate primer to bond to many commonly used thermoplastics. This not only eliminates the priming operation, but the cure cycles closely mimic the cooling cycle for the thermoplastic, allowing for in-mold bonding of the LSR to the thermoplastic. This is similar to overmolding of hard and soft thermoplastics, but the major difference is that the LSR side of the mold requires heat to crosslink. This should be taken into account when choosing the thermoplastic substrate, which must withstand the heat for curing the LSR without deformation. In some cases, residual heat in the solidified thermoplastic actually contributes to curing the LSR.
The ability to mate LSR to various thermoplastics without a priming step provides advantages in part design. LSR can be added only in the precise areas where it is needed for functional purposes, without any need to cover additional surface area just to hold the LSR in place. Likewise, areas of the thermoplastic substrate not overmolded with LSR no longer need added thickness to withstand the heat of curing the rubber. These factors can result in a more economical part based on using only as much of each material as needed. It can also result in smaller parts overall, contributing to the trend to miniaturization.
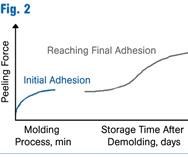
It is important to point out that current self-adhesive LSR technology is designed to provide only enough initial bond strength for part removal (no one wants the LSR to bond to the metal of the mold). The overall bond strength continues to increase over a couple of weeks, normally well before arriving at the end user (Fig. 2). If quicker maximum bond strength is required, this can be obtained with a 1-hr post-bake in an oven at 100 C/212 F.
About the Author
Gregory Dull is marketing manager for rubber fabricators at the Wacker Silicones unit of Wacker Chemical Corp., Adrian, Mich. (wacker.com). He can be reached at (517) 264-8271 or gregory.dull@wacker.com.
Related Content
Custom Molder Manages Growth on Several Fronts
Adding people, plants and machines, expanding capabilities in LSR, high-tonnage presses, automation and 3D printing—EVCO Plastics maintains momentum through challenging times.
Read MoreFakuma 2023: Wittmann Battenfeld Expands All-Electric Line, Direct-Current Capabilities
Wittmann Battenfeld will introduce the new EcoPower B8X injection molding machine line and show direct current as an energy source for a concept machine that will power its own robot.
Read MoreMinnesota Rubber and Plastics Opens Innovation Center
The plastics and rubber compounder and molder has opened the 9000-ft2 center that’s intended to help speed product development with design, prototyping, testing and production capabilities.
Read MoreSilicone Elastomer Course Returns
After completely filling the classroom last year, the Silicone Elastomers Technology and Fabrication course returns to Pomona, Calif. Jan. 31-Feb. 3.
Read MoreRead Next
LSR Part III-- Choosing a Mixing/Metering System
Liquid silicone rubber (LSR ) is a two component reactive chemical with a viscous, paste-like consistency.
Read MoreGetting Into LSR: Part II--Chosing an Injection Machine
The criteria for selecting a liquid silicone rubber injection molding machine are very similar to selecting a machine for standard thermoplastic injection molding, with several key differences.
Read More
















.png;maxWidth=300;quality=90)











