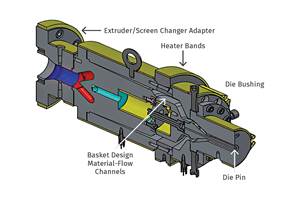
On Site: Growth Through Innovation in Medical Tubing
By focusing on high-tech, quick-turnaround, turnkey solutions, Putnam Plastics has grown into a leader in the field of medical extrusion.
Putnam Plastics was started in 1984 by Jim Dandeneau with the goal of being an innovator in medical tubing. A handful of employees, a couple of extruders, considerable tooling know-how, and various proprietary techniques—including a patented process to transition from a rigid to flexible material within a single-catheter construction—comprised the small custom processing operation, originally based in Danielson, Conn.
The firm has changed hands a couple of times since then. In 2004, it was purchased by Memry Corp., Bethel, Conn., to complement its metals-fabrication business for medical applications. Memry was bought by Italian metals company SAES Getters S.p.A. in 2008, paving the way for Dandeneau and business partner Lawrence Acquarulo to buy back Putnam Plastics in 2009.
Today, under the umbrella of the PolyMedex Discovery Group, Putnam Plastics employs about 150 people full-time and operates three shifts, five days a week, in a 40,000-ft² facility in Dayville, Conn. Expansion plans are on the horizon. The state-of-the-art operation (putnamplastics.com) is ISO 13485:2003 and 9001:2008 certified. It houses a fully equipped laboratory for incoming materials inspection and developing extrusions for challenging, minimally invasive applications. It is also used to scale up new processes for transition to manufacturing.
“Virtually 100% of our business is medical,” says Dandeneau. “We’re specialists in high-tech extrusion. Our business requires strong engineering. We do everything we can to control the process: in-house tooling, quality and process monitoring, continuous improvement, design for assemblies, a wide range of secondary operations. The more we do, the better our competitive position.”
Serving an industry where speed-to-market often rules the day, Putnam has brought in-house virtually every aspect of catheter production. It now considers itself a medium-sized operation within the specialty medical tubing field, but its control over the process and its involvement in front-end product development helps make Putnam as fast and nimble as smaller companies in the field.
“In medical, it’s all about speed,” Dandeneau remarks. “Medical-device companies are asking for protypes from processors in two weeks, from the time we download a client’s drawing with our SolidWorks 3D CAD software, through MasterCam software for programming our CNC machines to cut tooling, to extruding tubes. This is not an easy task with the current complexity of devices, yet we continue to focus on lean initiatives that reduce our lead times in line with the market needs.”
Medical is also about innovation. Business conditions in this market are influenced less by fluctuations in the overall economy, Dandeneau explains, and more by product lifecycles. “There are instances where tubing we made 20 years ago is still being used, but that is the exception rather than the norm,” he says. “It’s critical to be in on the next platform, to look for new solutions, to look for the next compound that perhaps processes better than what you are running now, to be technology-focused, to continuously improve your operation. We’re a medical extrusion technology leader, so we are prepared for any challenge that comes our way.”
Putnam has also stretched its wings to compete globally. Today, Dandeneau estimates that 30% of its business is international, most of that in Europe. This expansion comes at a time when more and more medical-device OEMs are moving away from having devices supplied exclusively by a single processor. “That presents an opportunity for us to gain business from OEMs who might have used our competitors exclusively,” states Dandeneau. “Of course it is a double-edge sword, as we could lose some business that we currently hold exclusively. Competing globally can help us offset some of that risk.”
TURNKEY SOLUTIONS
Consistent with its strategy, Putnam last year launched a custom components unit to focus primarily on post-processing steps such as printing and forming of catheters. Located in the Dayville plant, the operation includes fabricating catheter ends and access openings, as well as joining (including various types of welding and insert molding hubs and connectors), and printing of extruded tubes for intravascular and natural-orifice procedures. Traditionally, the device manufacturers themselves perform these finishing steps, or outsource them to third parties specializing in printing, assembling, or other technologies. Often, multiple third parties were required before a catheter extrusion was ready for final assembly of the device.
Putnam’s approach houses diverse operations specifically designed for catheter components—such as printing, welding and overmolding—under one roof. The custom components operation specializes in custom printing on catheter tubing and has expertise in 360° printing around the circumference of the catheter shaft and along the length.
“Printing is essential to catheters and a natural extension of the services we have traditionally offered device manufacturers,” says Dennis Cherok, director of operations for catheter components. “Our printing capabilities allow device manufacturers a one-stop-shop for extrusions finished to their final form with branding and functional markings.”
Putnam’s customized pad-printing equipment can print multiple colors on components over a meter long and apply graphics to the full outer circumference of the tubing. Advanced surface pretreatment technologies allow for excellent ink adhesion to even the most challenging polymers such as polyethylene, PEEK, and thermoset polyimide.
DEDICATED CELLS
A long way from its two-extruder origins in Danielson, Putnam now runs 13 extrusion lines using 38 single-screw extruders ranging from 0.5- to 2-in. diam. Primary equipment suppliers include Davis-Standard and American Kuhne. Putnam can run virtually all melt-processable thermoplastics—TPU, other TPEs (notably Arkema’s Pebax), nylon, PEEK, polycarbonate, EVA, PVC, polyolefins, acetal, polyesters, fluoropolymers, and more. The firm is moving more toward dedicated cells, where manufacturing lines are restricted to certain products.
Many of Putnam’s extrusion lines are equipped with gear pumps, which Dandeneau believes offer more precise control of melt flow to the tooling. On-line gauges and controls provide an additional control element. Putnam takes material drying particularly seriously. Depending on the resin, Putnam will dry materials centrally and then a second time with machine-mounted units, some of which are supplied by Dri-Air Industries. Putnam deploys both vacuum and water-tank cooling technologies—depending on the application—with equipment from the likes of Conair, RDN Manufacturing, and CDS. Products are cut either in-line or off-line, depending on length and other requirements.
From the beginning, Putnam has specialized in challenging extrusions, including tubing made of multiple materials and highly complex multi-lumen devices. To date it has produced tubes with up to 30 lumens. Its multi-lumen technology is available in diameters from 0.010 in. to 1 in., from round to complex profile shapes inside and out.
About 70% of the tubing Putnam processes are coextrusions. These include catheters requiring a portion be radiopaque under fluoroscopy, and a portion to be clear for visibility of the lumens. Other combinations are a hard, lubricious inner surface and soft, bondable outer surface for use in guide-wire catheters.
Back in the late 1980s, Putnam patented a process called Total Intermittent Extrusion. Today, it has a line dedicated to this technology, which can produce a single tube with variable durometer along its length to provide both flexibility and torque control. This eliminates the need to bond two different tubes to achieve the same result. Most of these tubes are TPU-based.
Putnam also has lines dedicated to wire and fiber jacketing. Longitudinal wires or fibers are incorporated into an extrusion to offer structural support or electrical data transmission. Reinforcements applied to single-lumen, profile, and multi-lumen extrusions include coils to resist collapse and buckling, and braid to improve burst strength and torque transmission. Reinforcing materials include stainless steel, nickel/titanium alloy, or high-tensile polymer fibers.
Putnam has recently made medical tubing of thermoset polyimide a more significant part of its product mix. “The mechanical, thermal, chemical, and radiation properties and tight tolerances of thermoset polyimide tubing are unmatched by any other material,” states Dandeneau. Polyimide tubing is available in a variety of colors and can include fluoropolymer additives for improved chemical resistance and lubricity.
While pure polyimide tubing is available only in round, single-lumen configurations, one or more polyimide tubes may be incorporated via proprietary techniques into a multi-lumen thermoplastic extrusion as a lumen lining. Diameters range from 0.007 to 0.080 in. with wall thicknesses from 0.0002 to 0.010 in. Like thermoplastics, thermoset polyimide tubing can be reinforced with stainless-steel wires in coil and/or braid configurations.
The Putnam plant is also equipped with a large walk-in annealing oven capable of applying customized heat cycles to extruded components up to 2 meters long. Annealing helps provide a dimensionally stable finished product.
While clean rooms are not required in Putnam’s business, it does have two white rooms equipped with two extruders for running tacky, dust-sensitive materials.
RECENT INNOVATIONS
As we reported in our December issue’s Starting Up section, Putnam recently developed a multi-layer tubing composed of thermoset polyimide and TPE. This teams the high strength and tight tolerances of the thermoset with the wide variety of material options and configurations available in TPE. Dandeneau says the coextrusion is ideal for demanding medical device applications that require a combination of high performance and manufacturability.
More recently, Putnam developed a new type of medical tubing said to be a first in combining multi-lumens and braid reinforcement. Previous endoscopic devices consisted of a large, coil-reinforced single-lumen extrusion with numerous smaller single lumens assembled within to provide working channels.
The former approach reduced the amount of available working space due to the wall thicknesses of each single extrusion and made the final assembly of the device much more complex and therefore more costly. The new coil-reinforced, multi-lumen tubing consolidates these independent extrusions, thereby increasing working space while reducing assembly time and cost, Dandeneau says. Reinforcements are available in coil or braid form, depending on therapeutic procedure requirements.
Related Content
Is Your Die Flow Changing Despite Following All the Correct Formulas?
Maybe the problem is that you're starting up with a dry die. Here are tips to solve this issue.
Read MoreBelling Machine Forms 'Rieber' Socket in PVC-O Pipe
Italy’s SICA has developed an innovative Rieber belling process specific for PVC-O pipes. At the same operating pressure, these pipes require a wall thickness of about 35-40% less than those in conventional PVC pipe and allow the construction of water supply pipes up to 25 bar of operating pressure.
Read MoreHow to Select the Right Tooling for Pipe Extrusion
In pipe extrusion, selecting or building a complementary set of tooling often poses challenges due to a range of qualitative factors. Here’s some guidance to help you out.
Read MoreWisconsin Firms Unite in Battle Against Covid
Teel Plastics opened new plant in record time, partnering with AEC & Aqua Poly Equipment Co. to expand production of swab sticks to fight pandemic.
Read MoreRead Next
Processor Turns to AI to Help Keep Machines Humming
At captive processor McConkey, a new generation of artificial intelligence models, highlighted by ChatGPT, is helping it wade through the shortage of skilled labor and keep its production lines churning out good parts.
Read MoreLead the Conversation, Change the Conversation
Coverage of single-use plastics can be both misleading and demoralizing. Here are 10 tips for changing the perception of the plastics industry at your company and in your community.
Read MoreAdvanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read More