Growing Interest in TPEs for COVID-19 Related Medical Applications
United Soft Plastics reports on a surging intereste for TPEs for face masks, ventilators, and face shields due to their elasticity, resilience, and FDA compliance.
Manufacturer and custom compounder of thermoplastic elastomers United Soft Plastics (USP), Lawrenceville, Ga., reports a surge of interest in the use of TPEs for the production of many COVID-19 related medical applications. The company says several companies, both domestic and international, have approached the company requesting materials to convert their non-medical manufacturing facilities to produce items to meet the healthcare demand from the global pandemic.
TPEs have been adopted in a range of COVID-19 related applications including personal protective equipment (PPE) like face masks and face shields, and also ventilator parts, according to USP’s executive v.p. and CFO Benedict Herbst. “We are pleased to be an important contributor in the development of medical products that will help our fellow citizens combat this awful virus.”
USP reports a spike in interest both in replacing current materials (i.e. silicone or standard rubber) with softer plastic in current applications and in scaling those applications for the future. “We see a larger fraction of consumers become more conscious of infectious disease and the ability that we have, both as manufacturers and consumers, to prevent the spread of disease through the choice of proper materials,” said Herbst. The result is that many applications which previously were not considered “medical” (i.e., a pen cup in a public waiting room) will in the future need to hold up to different sterilization methods such as autoclaving, according to Herbst.

USP, through its Mexican subsidiary USPM de Mexico Plastics, has supplied a special TPE compound for the Nvironone OvaOne interchangeable filter face mask from Mexican manufacturer Nvironone. The mask has three containment plates that progressively block the dispersion of droplets and utilizes an N95 filter that retains the spread of aerosol particles. The injection molded material for the mask’s base structure is designed to meet FDA contact requirements and is safe for skin contact. The high-performance TPE exhibits high tear strength and a smooth surface finish. This is just one example of custom developed TPEs for a range of face mask applications. “Despite the challenging business environment due to the pandemic, we’ve enjoyed strong growth in Mexico thanks to applications like these,” said Tomasz Nowinski, General Manager of USPM de Mexico.
Throughout the pandemic, there has also been strong growth in face shields and other face coverings. USP’s Unisoft Special and Medical Series grades are highly suitable because they provide excellent elasticity, strong recovery properties, and tear resistance for all elastic parts. To date, there have been several million face shields manufactured utilizing USP materials for the straps.
In response to continued growth, USP has undertaken extensive product development work and now offers a broad portfolio of TPE products. The company’s product line includes standard grades adhering to PP and special adhesion grades for non-PP overmolding applications with substrates such as ABS, PC, nylon, PBT, PS, PPO, and PMMA. Other innovative materials have been developed to meet special regulatory requirements within the FDA, EU food contact, REACH, and NSF. The range of products also includes new acrylic-based TPEs which offer an ultra-transparent profile and a smooth dry surface with improved oil and chemical resistance.
USP expects this demand to drop gradually towards the middle or end of 2021, but there will still be more long-term support for these medical products than existed before the pandemic, particularly due to the growing use of antimicrobial, antivirus, and antibiotic compounds.
Related Content
Resins & Additives for Sustainability in Vehicles, Electronics, Packaging & Medical
Material suppliers have been stepping up with resins and additives for the ‘circular economy,’ ranging from mechanically or chemically recycled to biobased content.
Read MoreTracing the History of Polymeric Materials -- Part 29: Polyurethane
This material family has unparalleled versatility, not only in terms of the forms the material can take, but in the different ways in which it can be processed.
Read More“Smart Nanocomposites” for Vehicle, Wearable Electronics and Buildings
OCSiAl’s graphene nanotubes combined with a TPU to fabricate nanocomposite sheets that can harvest electrical energy from sources of waste heat.
Read MoreCustom 3D Printed Parts Made From Bio-based Elastomers
Erpro's custom part printing service will now offer Arkema's Pebax RNew elastomer.
Read MoreRead Next
Why (and What) You Need to Dry
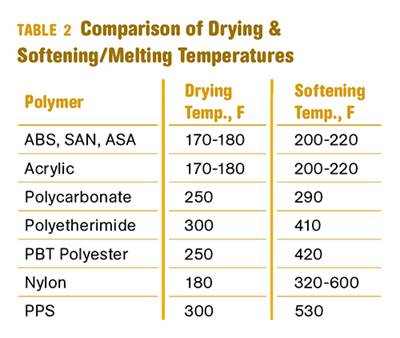
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreHow Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read MorePeople 4.0 – How to Get Buy-In from Your Staff for Industry 4.0 Systems
Implementing a production monitoring system as the foundation of a ‘smart factory’ is about integrating people with new technology as much as it is about integrating machines and computers. Here are tips from a company that has gone through the process.
Read More










.png;maxWidth=300;quality=90)