Cell Automates Overmolding, Inspection of Hypodermic Needles
Moldworx designed a mold, end-of-arm-tooling and automation cell to automate production of hypodermic needles, replacing manual post-mold gluing.
Asked by a medical OEM to develop a single-cavity injection mold to overmold a needle that was being manually glued in place, Moldworx LLC, a custom molder/moldmaker based in Gilbert, Ariz., did one better—or technically four better—developing a fully automated four-cavity tool and cell that runs on a horizontal press. The company initially proved out the concept on a single-cavity tool featuring slides where the needle was hand loaded. Jim Taylor, Moldworx president, told Plastics Technology that the new four-cavity automated cell runs on a 27-second cycle, matching the time used by the single-cavity tool but quadrupling the output. Before Moldworx developed the cell, the customer had three shifts of people gluing needles in place.
Taylor said that to shorten the lead time, Moldworx designed and built the entire automation cell in parallel with the mold. One challenge Moldworx had to overcome was singulating thousands of delicate, tiny needles—as small as 0.012 inch (0.305 mm)—that are bulk-packed. Moldworx ultimately designed and developed a “singulator” with a “hopper” that would introduce one needle at a time to the assembly line cell.
Each needle is picked up by a robot affixed to the injection molding machine and indexed in front of a set of high resolution inspection cameras to ensure it is not bent or that the needle tip is not damaged. The needles also must be oriented correctly, as there is a sharp end and a blunt end. Taylor said the system will discard the needle if the blunt end is up, trying this three times before alarming.
The needle is then placed into the tool for overmolding. Using gravity and a vertical press can often ease insert overmolding, but Moldworx designed this system to run on a horizontal press. To hold the needle in place prior to injection, Taylor said the tool features a slide with a core pin that has a .013Ø hole in it to accept the needle. The slide/core pin act as guides to allow the assembly to move away from the parting line and provide easier access to load the needle.
The process starts when the custom designed end of arm tool (EOAT), which is affixed to a 3-axis Yushin Robot, docks at the needle pick-up location and transfers the needle to the mold, docking with the tool to ensure precise positioning. In its first action, the EOAT grips the finished part and retracts to move it out of the way.
Once a sensor verifies the EOAT has the part, it moves towards the slide and engages with the guide pins. At this point, the gripper with the needle is just above the slide core pin. A servo cylinder on the slide moves the core pin up to allow the needle to move into the hole; the servo moves to the finished install position; and the EOAT reverses its sequence. A laser installed in the mold verifies the needle is present before the molding cycle begins.
When the mold opens, the robot removes the molded assembly and places a newly inspected needle into the mold. Moldworx noted that all these actions require inspection and verification, and tolerances must be exact.
After molding, the end of arm brings the finished, molded assembly to the off-load position in the assembly cell, according to Taylor, placing the molded part on a mandrel before leaving to get another needle. At this station, two cameras verify the sharp end is up; the tip is not damaged; and the needle is not bent. If the needle passes inspection, the table indexes to the next position where filtered, metered air is pushed thru the needle to check for occlusion.Related Content
Allegheny Performance Plastics to Enter Health Care Sector
The custom molder has secured multiple projects in health care and will be adding cleanroom and white-room spaces, as well as injection molding machines, in support.
Read MoreUse Cavity Pressure Measurement to Simplify GMP-Compliant Medical Molding
Cavity-pressure monitoring describes precisely what’s taking place inside the mold, providing a transparent view of the conditions under which a part is created and ensuring conformance with GMP and ISO 13485 in medical injection molding.
Read MoreSeaway Plastics Acquired
Private equity firm ICG purchased the injection molder and its three facilities located in Florida and California.
Read MoreWisconsin Firms Unite in Battle Against Covid
Teel Plastics opened new plant in record time, partnering with AEC & Aqua Poly Equipment Co. to expand production of swab sticks to fight pandemic.
Read MoreRead Next
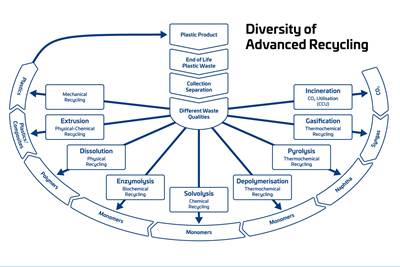
Advanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read MoreWhy (and What) You Need to Dry
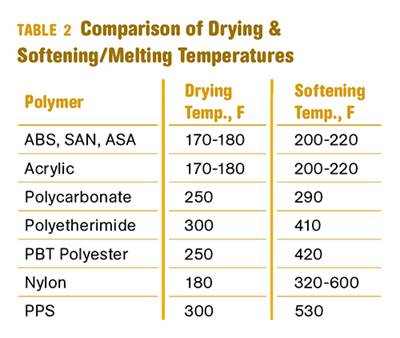
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreUnderstanding Melting in Single-Screw Extruders
You can better visualize the melting process by “flipping” the observation point so that the barrel appears to be turning clockwise around a stationary screw.
Read More