Pre-Colored Medical ABS Compounds for Laser Marking
Elix Polymers’ specialty medical pre-colored ABS compounds are based on existing biocompatible M203FC and M205FC.
Specialty medical pre-colored ABS compounds from Spain’s Elix Polymers (U.S. office in Weston, Fla.) have been optimized for laser marking of the mandatory unique device identification (UDI) that is required by actual medical legislations to protect against counterfeiting. EU’s new Medical Device Regulation (MDR) and US’ Code of Federal Regulations Title 21 on labelling of medical devices (21 CFR 801 Subpart B) enforce the use of a UDI system to be placed on medical devices and on their packaging to reduce the risk of counterfeiting, improve traceability and better manage incident reporting, device recalls and other adverse events.
Several medical devices now must be directly marked with a unique device identifier (CFR 21 Sec. 801.45 or EU MDR Part C 4.10 about the UDI system in reusable devices); in general, if the devices are intended to be used more than once and to be cleaned or disinfected before each use. Laser marking makes it possible to attach a unique identifier to the medical device and offers a series of additional advantages compared to alternative technologies such as labelling, pad printing, or hot stamping. Some materials can be more easily laser-marked than others, while some will provide very poor results. Special attention also should be given to the material’s color; in particular, the color formulation and its compatibility with the base material to create certain laser marking effects or colors of contrast.

ABS compounds are now widely used to produce external enclosures, covers or shells for reusable devices. ABS is a laser markable material where correct laser settings adapted to the ABS material produce acceptable laser marking results. However, there may be some limitations since the text or information to be laser-marked could produce an undesired color, level of contrast or an unexpected resolution. Additionally, there is an important risk of no longer complying with regulations, especially in the medical devices sector. For these reasons Elix Polymers has developed specialty medical ABS formulations based on M203FC and M205FC materials that optimize laser marking to achieve a specific color change and a targeted level of contrast over a given color background that is fully compliant with regulations.
According to Elix, it specialty ABS formulations are also particularly valuable when other relevant material properties must be maintained along with laser marking optimization, such as the biocompatibility requirement of ISO 10993. Since ABS and its color are both important factors for obtaining the required laser marking effect, a critical aspect of this type of development is to create a database selection of available pigments with the highest performance under different laser systems.
In the case of medical devices requiring biocompatibility, among the available pigments used for optimizing laser marking contrast, only the ones with biocompatibility properties and within the legal admissible concentrations can be used to guarantee compliance with the required medical regulation. Elix’s Product Stewardship department is said to provide essential support for this task, which also adds value by conducting a constant surveillance of admitted substances and concentrations and by guaranteeing that the complete formulation, including the color formulation, stay within legal limits. Recombined validated pigments provide new color formulations for medical ABS, where laser marking contrast is enhanced while guaranteeing the same target color material is maintained.
Related Content
The Fantasy and Reality of Raw Material Shelf Life: Part 1
Is a two-year-old hygroscopic resin kept in its original packaging still useful? Let’s try to answer that question and clear up some misconceptions.
Read MoreWhat's the Allowable Moisture Content in Nylons? It Depends: Part 2
Operating within guidelines from material suppliers can produce levels of polymer degradation. Get around it with better control over either the temperature of the melt or the barrel residence time.
Read MoreVolume Resin Prices Move in Different Directions
PE, PP, PVC, and ABS prices slump, while PS, PET, PC, and nylons 6 and 66 prices rise.
Read MoreTracing the History of Polymeric Materials: Aliphatic Polyketone
Aliphatic polyketone is a material that gets little attention but is similar in chemistry to nylons, polyesters and acetals.
Read MoreRead Next
How Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read MoreWhy (and What) You Need to Dry
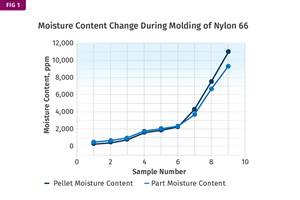
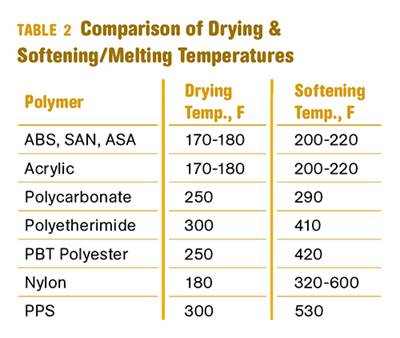
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreLead the Conversation, Change the Conversation
Coverage of single-use plastics can be both misleading and demoralizing. Here are 10 tips for changing the perception of the plastics industry at your company and in your community.
Read More