Modifying PVC with Bio-Based PHA Rubber
A new class of renewable and very efficient multifunctional modifiers for PVC has been developed at Metabolix. This series of biobased PHA (polyhydroxyalkanoate) copolymers offers toughening, plasticization, and improved processing of all types of PVC compounds. They also have good UV stability and transparency, do not promote biodegradation, and are fungi resistant.
A new class of renewable and very efficient multifunctional modifiers for PVC has been developed at Metabolix. This series of biobased PHA (polyhydroxyalkanoate) copolymers offers toughening, plasticization, and improved processing of all types of PVC compounds. They also have good UV stability and transparency, do not promote biodegradation, and are fungi resistant.
Because of their unique miscibility with PVC, the PHA modifiers do not migrate, extract, or volatilize, and they are easy to handle and process at the same conditions as PVC. They offer a new, simplified approach to PVC formulation in which multiple processing and modifying additives could be replaced with a single product, eliminating both undesirable additive interactions and the need to stabilize unstable ingredients.
The centerpiece of the Metabolix plant-based resin technology is PHB (polyhdroxybutyrate), the simplest member of the broad PHA family of biopolymers. Metabolix’s current range of Mirel PHA copolymers are produced by bio-fermentation of sugars in which the water-insoluble inert polymer accumulates inside specially engineered microorganisms. The polymer is then extracted and purified and can be compounded and processed using conventional plastics converting equipment.
In our recent study, we used three different PHA copolymers as modifiers for PVC: I6001, a semi-crystalline PHB copolymer with a nominal crystallinity of about 25% and glass-transition temperature (Tg) of about -8 C; I6002, an amorphous PHB copolymer with a Tg of about -21 C; and a developmental, next-generation PHA grade for rigid PVC. The PHA copolymers can contain hard crystalline and soft rubbery segments, which is very important for impact modification. Both phases have good miscibility with PVC.
PVC resin with a K-value of 70 was used for blending with PHA. For additional and comparative plasticization we used DIDP as the less extractable monomeric phthalate, as well as DINA (diisononyl adipate). For heat stabilization we used liquid BaZn carboxylates. The PVC/PHA blends are quite transparent when stabilized with liquid carboxylates. Commercially available impact modifiers of MBS, ABS, ASA, and CPE types were used for comparison.
TOUGHENING PLUS PROCESSING EASE
PVC has a well known propensity for brittle fracture, particularly in the presence of sharp notches or cracks and at low temperatures or high deformation rates. One way to enhance fracture toughness under these conditions is to incorporate a rubbery phase. Less polar elastomers, such as polybutadiene, natural rubber, butyl rubber, and styrene-butadiene rubbers are not useful for PVC modification due to their incompatibility and phase separation. The usual way to solve the incompatibility issues is to use grafted core-shell impact modifiers, such as MBS, AIM (all-acrylic impact modifiers), or ABS, whereby one phase—e.g., methyl methacrylate—is miscible with PVC and provides good adhesion with the matrix, and another phase—e.g., n-butyl acrylate or butadiene rubber—absorbs and dissipates the energy of an impact.
The performance of toughened PVC in this case depends strongly on the type of rubber and its dispersion quality, structure, size, and particle-size distribution. In addition, the rubber’s glass-transition temperature (Tg) must be lower than the intended service temperature of the PVC compound, and the rubber particles must be thermally stable at the processing conditions.
Metabolix’s newly developed PHA copolymer “rubbers” are miscible with PVC, have a Tg range from -8 to -28 C, and are thermally stable at PVC processing conditions.
It is generally accepted that a blend with a pseudo-network morphology has a higher toughening efficiency than a blend with a morphology of well-dispersed particles. At Metabolix, we have tried using this approach to create PHA-based toughening modifiers for PVC. Our expectation was that the entangled PVC/PHA networks would be less sensitive to processing, as they would not require the high-shear conditions needed to produce the required melt viscosity for the core-shell particle dispersion.
Along with their ability to promote shear melting of PVC, PHA modifiers also reduce the fluxing time and improve the overall thermal stability of PVC. We used a typical PVC two-roll mill for compounding at 330 F (165 C). The PHA copolymers added at up to 40 phr presented no processing problems. In fact, all PVC/PHA rigid and semi-rigid blends released nicely from the rolls and promoted much easier fusion of PVC particles than with other toughening additives, especially of CPE and MBS types. All the tested impact modifiers (CPE, MBS, ASA, and ABS) took two to five times longer to properly melt PVC resin than with PHA and required additional processing aids. Due to their wall-adhering nature and miscibility with PVC, PHA polymers promote shear melting, reduce the processing time, and do not require processing aids.
The milled sheet was then compression molded at 340 F into testing samples for tensile, flexural, and notched Izod impact performance, as well as Shore D hardness, and Tg (measured using a Dynamic Mechanical Analyzer).
PLASTICIZATION ADVANTAGES
PVC is plasticized to achieve flexibility, but the introduction of plasticizers results in changes to practically every other property. Impact strength and tensile toughness, for instance, are typically reduced with plasticization.
Miscibility between PVC and plasticizer is essential for good performance. If the plasticizer is not fully miscible and has a low molecular weight, it will migrate to the surface of the PVC article and form a separate layer. It will also be extractable, responsible for high volatile loss, staining, and lost properties upon aging.
Polymeric plasticizers can eliminate some of these deficiencies, but there are only a few commercially important high-molecular-weight plasticizers and they are quite inefficient, expensive, and difficult to homogenize into compounded PVC.
Metabolix has been able to demonstrate that its new PVC-miscible PHA copolymers act as very efficient, non-extractable, non-volatile polymeric plasticizers (at least up to 40 phr of PHA), and are environmentally safe and non-toxic. (Mirel grade I6001 also has FDA food-contact clearance per FCN 001119, which allows its use in contact with all foods except those containing alcohol under Conditions of Use B through H.)
Based on the Van Krevelen solubility parameters, the polar and nonpolar group contributions in PHA and PVC are very similar and miscibility could be expected when the chains have sufficient energy and similar mobility. In fact, there is no molecule with closer solubility parameters to PVC than the PHAs used in this study—including PMMA, the most miscible polymer used in the majority of core-shell impact modifiers.
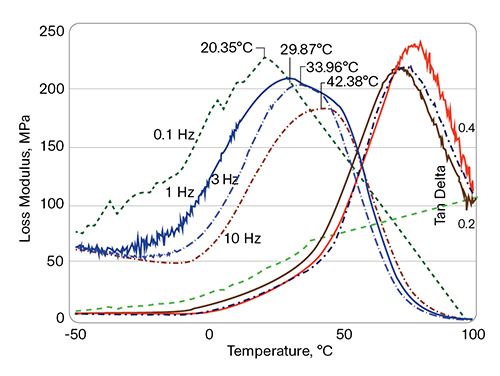
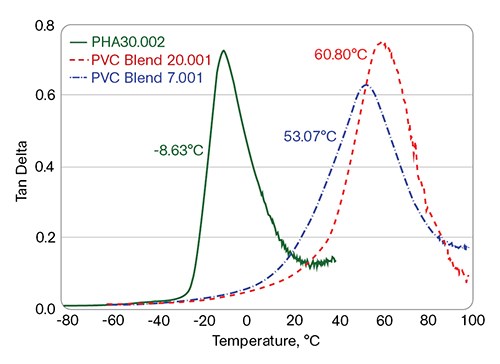
The multi-frequency DMA of the PVC/PHA blends (see Fig. 1) shows a single Tg, which also points directly to the blends’ miscibility. In the semi-rigid PVC compounds (all plasticized with 18 phr of DIDP), the PVC/PHA blends are also shown to be miscible: A single Tg is observed for the PVC/10 phr I6002 blend, which is shifted to a lower temperature than the PVC control (Fig. 2).
ENHANCING SEMI-RIGID & FLEXIBLE PVC
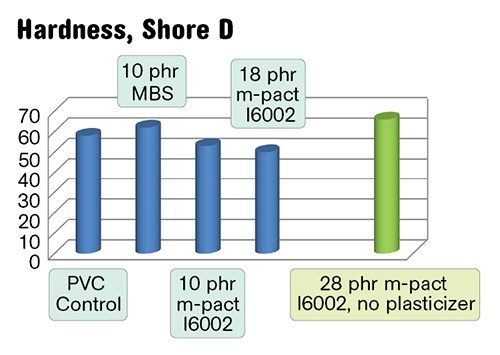
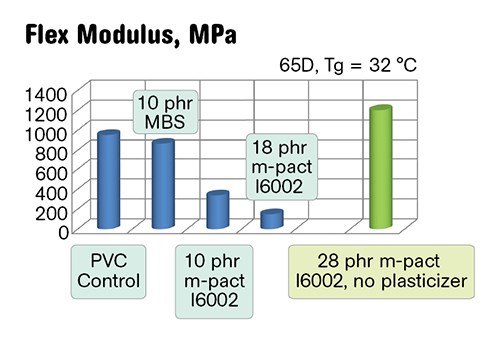
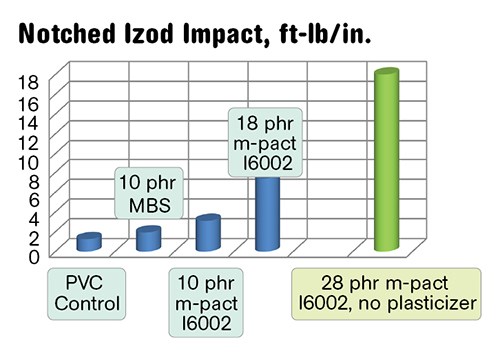
Physical properties of the semi-rigid PVC/PHA blends and a semi-rigid control are shown in Figs. 3a-c. The blends with higher impact also demonstrate higher flexibility due to the simultaneous plasticization. In green, we have shown a PVC/28 phr I6002 blend that contains no primary plasticizer. This compound has 65 Shore D hardness and reduced Tg and is very tough and impact resistant.
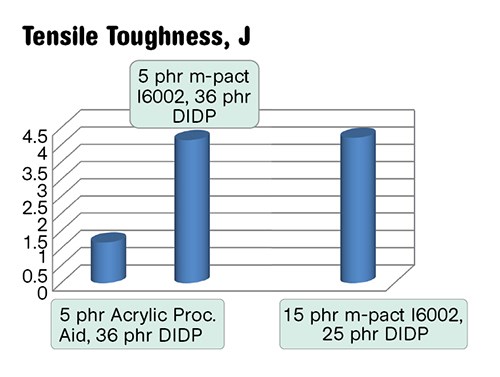
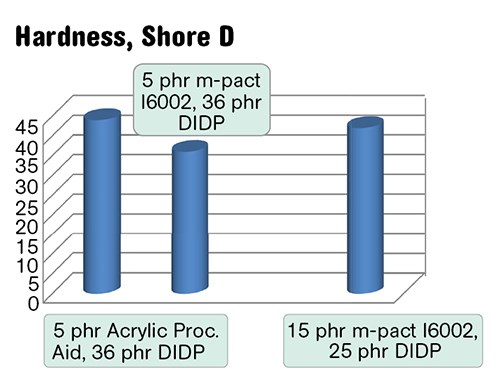
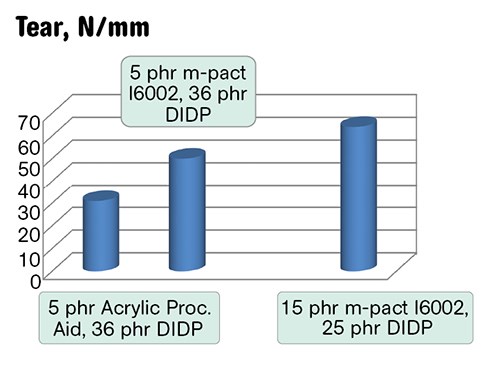
In flexible compounds with 25 to 35 phr of primary plasticizer, PHAs improve tear resistance, tensile toughness, and provide plasticization (Figs. 4a-c). The plasticization by a miscible polymeric additive that is non-extractable, non-migrating, and containing no volatiles or toxic chemicals should be especially valuable in medical, electric, automotive, packaging, and long-lasting applications. The ratios of primary plasticizer to PHA could be adjusted to match required properties. For example, as shown in Figs. 4b-c, a compound with a similar hardness could be made much more tear resistant if the primary plasticizer/PHA ratio changed from 36/5 to 25/15 phr.
BOOSTING RIGID PVC PROPERTIES
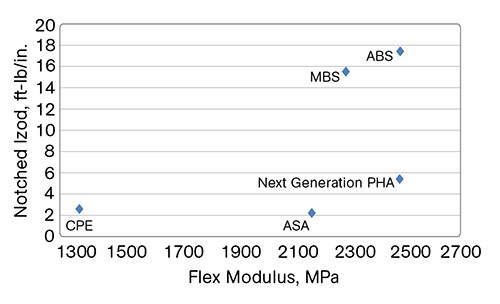
Based on the above data, both I6001 and I6002 PHAs provide plasticization and flexibilization of PVC. In rigid PVC compounds where a balance of toughness and stiffness is required, another type of PHA is needed. For impact performance, the toughening rubber segments should form separate small domains and the interfacial adhesion should be provided by sufficient miscibility with PVC segments. While these products are still being optimized and not commercial, the best-performing “next-generation” PHA modifier is mapped against most popular rigid impact modifiers in Fig. 5.
As shown, the impact modifiers with the best toughness and stiffness are MBS and ABS. These modifiers, however, cannot be used in compounds for outdoor use due to their oxidation and poor light stability. The impact modifiers developed for outdoor use, CPE and ASA types, have much worse impact strength and are much more flexible. The next-generation PHA confers much higher stiffness than do CPE and ASA modifiers, as well as better notched impact and very good light stability. It could be used in transparent applications.
LONG-TERM UV STABILITY
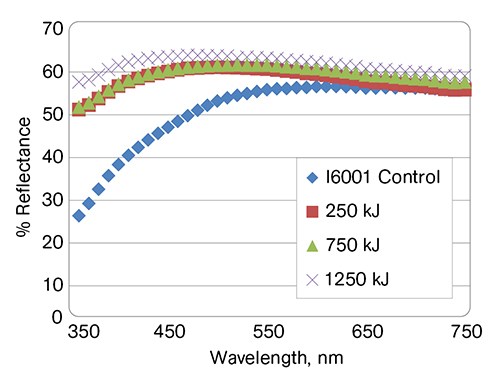
UV aging of the semi-rigid blends with 18 phr of DINA was conducted according to ASTM G155-2005 in a Xenon Ci65 weatherometer. Because they are linear aliphatic polyesters, PHA polymers do not absorb UV radiation and are known to have good light stability. The Metabolix PHAs, however, contain some traces of biomass, which tend to bleach off with UV exposure. Figure 6 shows the changes in reflectance with UV exposure for the I6001 sample: A gradual increase in the blue reflectance and lightness could be observed. This behavior is very similar to that of optical brighteners, which absorb UV light and re-emit it at the blue end of the visible spectrum. A photograph demonstrating the progress of bleaching of I6001 PHA is shown in the top left row of Fig. 7.
As a modifier, PHA offsets UV-induced yellowing of PVC and provides a very good color hold throughout the entire length of UV exposure, as shown in the lower left row of Fig. 7 and in Fig. 8.
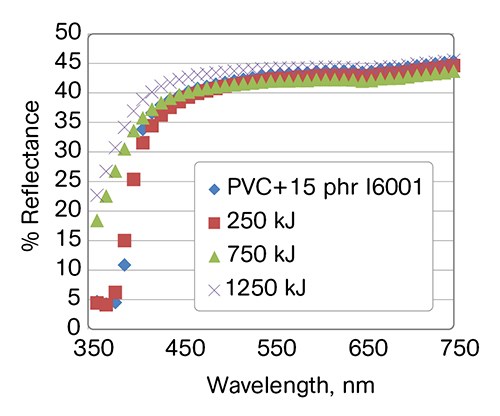
For comparison in Fig. 7, we have included compounds where PHA was replaced by the same levels of ASA and ABS impact modifiers. None of the compounds contain UV stabilizers and otherwise have identical composition (PVC, 18 phr DINA plasticizer, 4.5 phr ESO, 4 phr BaZn liquid stabilizer, 15 phr impact modifier). It is apparent that the semi-rigid compound containing I6001 PHA has the best UV stability and probably does not need a UV stabilizer. We are continuing to test the mechanical properties to confirm this finding.
FUNGAL RESISTANCE & SOIL BIODEGRADATION
The fungal resistance of PVC/PHA blends was tested according to ASTM G21-09 (28 days at 28-30 C and >85% relative humidity; 5 active organisms). The PVC control formulations contained 4 phr BaZn liquid heat stabilizer and 4.5 phr ESO. One of our three samples demonstrated traces of fungal growth. When I6001 was added to the control compound at 15 phr, all three PVC/PHA samples demonstrated no traces of fungal growth.
PHA polymers are known to be biodegradable. However, when used as additives in compounds with non-biodegradable polymers as a major phase, they do not promote biodegradation. We ran a soil disintegration test using Massachusetts farmland and maintained the moisture level at 10-12% at 20-23 C. The I6001 control lost 36% of its weight after eight weeks of soil burial, while its blends with PVC (both unplasticized and with 18 phr DINA) demonstrated no weight loss.
About the Author
Yelena Kann joined Metabolix, Inc., Cambridge, Mass., as a senior polymer scientist focusing on PHA technologies for polymer compounds, sheet, fibers, foam, and extrusion coating. She earned her Ph.D. in Chemistry in 2003 from the University of Sussex, U.K. Contact: (978) 513-1819; kann@metabolix.com; metabolix.com.
Related Content
Masterbatches Reduce Gloss in PLA and PETG 3D Printed Products
Insight Polymers & Compounding’s two low-gloss additive masterbatches shown to boost appearance of 3D printed objects.
Read MoreTeknor Apex Acquires Danimer Scientific
Danimer is a developer and producer of bioplastics.
Read MoreNPE2024 Materials: Spotlight on Sustainability with Performance
Across the show, sustainability ruled in new materials technology, from polyolefins and engineering resins to biobased materials.
Read MorePHA Compound Molded into “World’s First” Biodegradable Bottle Closures
Beyond Plastic and partners have created a certified biodegradable PHA compound that can be injection molded into 38-mm closures in a sub 6-second cycle from a multicavity hot runner tool.
Read MoreRead Next
For PLASTICS' CEO Seaholm, NPE to Shine Light on Sustainability Successes
With advocacy, communication and sustainability as three main pillars, Seaholm leads a trade association to NPE that ‘is more active today than we have ever been.’
Read MorePeople 4.0 – How to Get Buy-In from Your Staff for Industry 4.0 Systems
Implementing a production monitoring system as the foundation of a ‘smart factory’ is about integrating people with new technology as much as it is about integrating machines and computers. Here are tips from a company that has gone through the process.
Read More