Injection Molding PHA Bioplastics: Validating Moldability for Paper Mate Pens
As part of the commercialization process for Mirel PHA bioplastics, it became necessary to validate the injection molding process for a high-profile consumer products launch.
As part of the commercialization process for Mirel PHA bioplastics, it became necessary to validate the injection molding process for a high-profile consumer products launch. Since Mirel is a new polymer with different properties and processing characteristics, it is only natural to question the ability to use conventional tools to develop molded parts from Mirel. By partnering with a major consumer product manufacturer, we were able to explore the limits of what can be done. A new line of consumer products was designed and developed using Mirel PHA as the primary material. The process included using Autodesk Moldflow software to analyze the part filling and design of multi-cavity, hot-runner production tools. Through this process we were able to demonstrate that Mirel bioplastics can be molded similar to petroleum-based plastics.
WHAT IS MIREL?
Mirel is a family of bioplastic materials with physical properties comparable to petroleum-based resins. Mirel is both biobased and biodegradable in natural soil and water environments and in home and industrial composting systems. Mirel is a type of PHA (polyhydroxyalkanoate)— specifically, a proprietary blend of PHB (polyhydroxybutyrate) copolymers, additives, and mineral fillers. It is a type of semicrystalline thermoplastic polyester that is derived from microbial fermentation of corn sugar. The rate and extent of Mirel’s biodegradability will depend on the size and shape of the articles made from it. However, like nearly all bioplastics and organic matter, Mirel is not designed to biodegrade in conventional landfills.
Mirel is supplied by Telles (mirelplastics.com), a joint venture of Metabolix, Inc. and Archer Daniels Midland Co. The first commercial Mirel polymer plant came on stream late last year with a design capacity of 110 million lb/yr. Mirel is available for film and sheet extrusion and thermoforming, as well as injection and blow molding. Certain grades recently received FDA clearance for food-contact applications.
All Mirel bioplastics are thermally sensitive and must be dried to a low moisture content (0.1%). Their thermal sensitivity is a primary factor distinguishing proper molding technique for Mirel from that for other thermoplastics.
VALIDATING MOLDABILITY
Over the years many tools have been developed to help understand, model, and optimize both the design and processing of plastics parts. Since Mirel behaves differently from conventional plastics, it is only natural to question the applicability of these tools, developed for other plastics, with Mirel. Software such as Moldflow (moldflow.com) for analyzing part and mold design and the injection molding process can be a very powerful tool to accelerate the design and development process for parts produced from Mirel. Such tools also aid in making a robustly designed part, mold, and process.
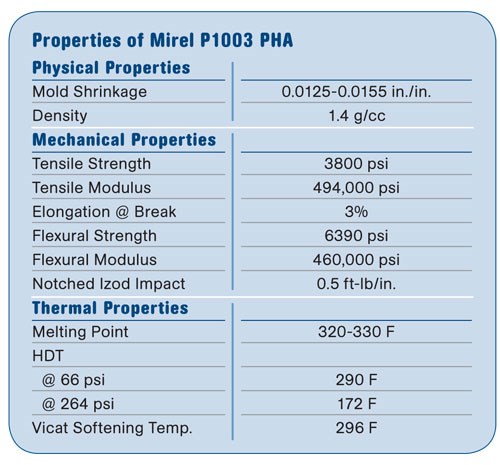
One of the most pressing questions was whether hot runners could be used with Mirel, because of its thermal sensitivity. Newell Rubbermaid’s Paper Mate div. (newellrubbermaid.com) had decided to produce a line of pens and mechanical pencils called Biodegradables from Mirel bioplastics. The product line was designed to have 60% biodegradable content from Mirel P1003 injection molding grade. This meant that the barrel, nosecone, and clip would have to be made from Mirel. Once Paper Mate had designed the components it was time to apply traditional engineering tools to analyze the processing of the individual components and to design commercial tools to make the components.
FLOW SIMULATION COMES FIRST
In order to design the molds for optimum processing and reduce the tooling development time, it was decided to perform Moldflow analysis of the designs before building the molds. The pen and mechanical pencil components were to be made from Mirel P1003, an improved injection grade introduced last year with enhanced stiffness and melt flow. In order to perform a Moldflow analysis, the material to be molded must have appropriate data in the Moldflow database. In order to simulate the injection molding process, Moldflow needs data on melt viscosity under different conditions of shear and temperature, melt density, melt thermal conductivity and heat capacity, and PVT data. Since Mirel P1003 is a new material, no such data existed yet. We would have to have this material analyzed by Moldflow. Analysis of Mirel P1003 was carried out by the Moldflow Plastics Lab in Ithaca, N.Y. A Moldflow database file for Mirel was created and subsequently used to perform the analysis of the proposed mold designs before the molds were built.
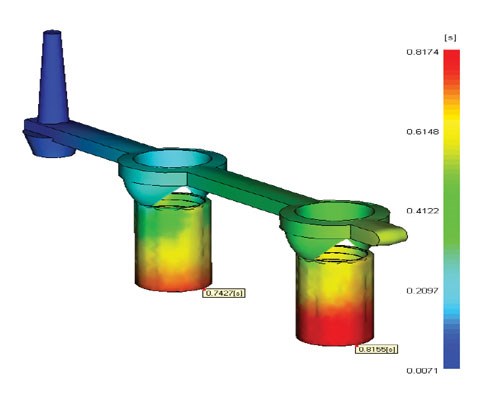
The analysis of the designs proved invaluable in accurately predicting troublespots in the design. These potential issues were identified based on the knowledge of how Mirel behaves in a mold. A principal example is the sensitivity that Mirel has to mold-filling imbalance. As with most plastics, a mold for Mirel must be designed with proper filling balance so that portions of a mold do not fill before others. In the case of Mirel, a small imbalance will lead to significant flash and/or short shots. This greatly increases scrap rates and forces a less than ideal injection molding cycle.
This program required multiple molds with four or eight cavities apiece. For the volume of parts to be molded, it was not economical to use single-cavity tools. The second reason for multicavity molds was to optimize shot size. With Mirel, it is important to maintain a short residence time in the barrel while molding. Too much time at elevated temperatures causes significant loss of molecular weight, which will ultimately lead to loss of mechanical properties. Mirel processes best when the barrel-capacity to shot-size ratio is between 2:1 and 4:1. This was another argument for multicavity molds.
The analysis in the accompanying Moldflow screen shot shows an example of a family mold with poor balance between cavities. This was the original proposal from the molder. Making family molds for Mirel is not a good option. The flow imbalance that results from the different cavity sizes causes significant scrap. A mold built with this design would result in almost 50% scrap due to either flash or short shots. This issue was avoided by redesigning the mold to be more geometrically balanced with multiple cavities of the same size. This meant moving the location of the thermal sprue between the cavities. It also meant using more thermal sprues than originally planned.
DESIGNING THE MOLD
The Moldflow analysis showed the best locations for gates and where end of fill would be. To reduce the amount of regrind and scrap, hot runners and thermal sprues with small cold runners were used for the different mold designs. The combination of thermal sprues with a small hot runner allows for a sub-gate design. This was necessary due to the size of the parts being molded. Smaller parts make it difficult to use full hot-runner systems due to the amount of space required for the hot runner. Using a full hot runner would make the overall dimensions of the mold too large to fit in the small injection molding machines being used for the project.
Husky Injection Molding Systems, Ltd. (husky.ca) designed the hot runners and thermal sprues to work with Mirel. After an in-house performance test with Mirel, Husky was able to recommend what style of hot runner and thermal sprue would work best with Mirel. The molders then designed each injection mold using the recommendations from Husky, the results of the Moldflow analysis, and the recommendations from the Mirel technical services team.
It is important that molds are designed with proper vents and gates. Mirel does not mold well with large vents or very small gates. Due to the viscosity profile of Mirel, large vents cause flash. It is recommended that vents for Mirel be 0.0005 in. deep. Small gates cause too much shear when molding, which tends to lengthen the overall injection molding cycle. In addition, the heat generation from gate shear causes shear thinning of Mirel resin, increasing the potential for flash. Since these molds were being designed and built specifically for Mirel, we were able to make sure that the proper design recommendations were incorporated into the design. (Complete injection mold design recommendations can be found at mirelplastics.com.)
INJECTION MOLDING GUIDELINES
The best designed and built molds will not run well if the processing is not right. Some key parameters for injection molding Mirel P1003 include the barrel temperature profile, mold temperature, screw speed, screw backpressure, and injection speed. By balancing these parameters it is possible to injection mold Mirel efficiently and economically.
Mirel must be molded using a reverse temperature profile. This means using a higher temperature in the rear barrel sections under the hopper than at the nozzle. A typical process profile would be 360, 350, 330, and 330 F from rear barrel zone to nozzle. The goal is to achieve a melt temperature of 330 to 340 F. The reason for the reverse profile is to control melting of the polymer and limit the amount of molecular-weight loss.
For the same reason, the screw speed and backpressure are limited as well. Screw speed and backpressure can contribute significantly to the amount of energy put into the polymer during melting. Typical screw speed for Mirel is 100 rpm and backpressure is typically 50 to 100 psi. A low-shear, general-purpose screw is recommended. It should have a short L/D and compression ratio of 2.2:1 to 2.6:1.
Mirel also responds best to a slow injection speed. This is, again, partly in order to minimize gate shear and shear heating. A short, reverse-taper nozzle is recommended.
Mirel has a slow crystallization rate. This allows for a longer injection time and better filling of complex geometries. The slow injection speed does not have a negative impact on the overall molding cycle as this time would normally be taken up with pack and hold, which can be shorter with slow injection.
The other critical process parameter for Mirel is the mold temperature. It should be controlled at 140 F. This is very important for Mirel because its fastest rate of crystallization occurs at 140 F. A small deviation from this will have a significant impact on the cooling time needed to eject parts from the mold. Cooling the mold below 140 F does not speed the crystallization rate and will increase the cooling time. As little as a 20° F deviation will double the cooling time needed.
With Mirel it is also necessary to monitor the quality of the regrind being used. Up to 20% Mirel regrind can be used in injection molding. Typically, regrind is comprised of startup scrap and runners. Since the molds for the Paper Mate Biodegradable pens and mechanical pencils were designed with hot runners and thermal sprues, the amount of runner regrind is kept to a minimum. The regrind must still be monitored in order to ensure that it is still good material and has not been degraded. It also must be properly dried to below 0.1% moisture. (That usually takes 2 to 4 hr at 176 F in a dessicant dryer with -40 F dewpoint. Parts that contain bubbles or stick to the mold may indicate excessive moisture in the material.) The amount of regrind should be kept constant as large fluctuation in the amount or quality of regrind will affect the injection molding cycle. (Detailed processing information can be found at mirelplastics.com.)
THE RESULTS
The molders have been operating the injection molds for the Paper Mate Biodegradable pens and mechanical pencils for several months with minimal issues. Efficiency continues to be improved in the overall molding process, and reduced cycle times have been achieved relative to traditional plastics. The molds are all running with minimal scrap—approximately 4%. We have been able to process several different lots of Mirel P1003 with minimal adjustment to the molding process. This, combined with the lower processing temperatures, results in significantly lower energy consumption to produce the Paper Mate Biodegradable pens and mechanical pencils from Mirel P1003 relative to conventional plastics. Measurements show a 20% reduction in electrical consumption in molding the mechanical pencils as compared with ABS or polycarbonate. Modeling indicates a similar reduction in energy for the pens.
Bret Marschand, senior development manager for Newell Rubbermaid Office Products conducted a thorough evaluation of various biobased materials for use in the company’s products. he selected Mirel for its unique combination of performance and biodegradability.
“Mirel molds very well, processes in conventional injection molding equipment, and is durable in use,” said Marschand. “Add these characteristics to its biodegradability profile, and we have found a remarkable material that fits into our global corporate sustainability goals.”
About the Author
Thomas J. Pitzi is manager of technical service and application development for Telles in Lowell, Mass. He has over 15 years’ experience in plastics as a technical service and application development engineer at firms such as Arkema and DSM Thermoplastic Elastomers. Pitzi can be reached at (978) 513-1865 or tpitzi@mirelplastics.com.
Related Content
Advanced Biobased Materials Company PlantSwitch Gets Support for Commercialization
With participation from venture investment firm NexPoint Capital, PlantSwitch closes it $8M bridge financing round.
Read MoreResins & Additives for Sustainability in Vehicles, Electronics, Packaging & Medical
Material suppliers have been stepping up with resins and additives for the ‘circular economy,’ ranging from mechanically or chemically recycled to biobased content.
Read MoreHow to Optimize Your Molds and Hot Runners for Processing Bioresins
Demand for bioresins is growing in molded goods, particularly as a sustainability play to replace fossil-fuel based materials, but these materials are not a drop-in replacement for traditional materials. Molds and hot runners need to be optimized for these materials.
Read MoreHow to Extrusion Blow Mold PHA/PLA Blends
You need to pay attention to the inherent characteristics of biopolymers PHA/PLA materials when setting process parameters to realize better and more consistent outcomes.
Read MoreRead Next
How Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read MoreAdvanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read MorePeople 4.0 – How to Get Buy-In from Your Staff for Industry 4.0 Systems
Implementing a production monitoring system as the foundation of a ‘smart factory’ is about integrating people with new technology as much as it is about integrating machines and computers. Here are tips from a company that has gone through the process.
Read More