The Importance of Melt & Mold Temperature
Molders should realize how significantly process conditions can influence the final properties of the part.

As I work with processors I find a general lack of appreciation for how significantly process conditions can influence the final properties of the molded part. The prevailing opinion appears to be that the selected material exhibits the properties published on the data sheet, independent of how the raw material is converted into the molded article.
Under this way of thinking, the processor’s job is simply to heat the material to the molten state, pass it through the appropriate piece of processing equipment, and re-solidify the polymer into the shape described by the print. As long as the part fulfills aesthetic expectations and the critical dimensions meet the print, the processor has done his job. Properties are the province of the material supplier.
Unfortunately, it is not that simple. In injection molding, for example, molding conditions have a significant influence on the final properties of the material regardless of the part design. Two of the process conditions that have a substantial influence on the behavior of the polymer are the melt temperature and mold temperature.
First, it is important to distinguish between these process conditions and the setpoints that we use to exercise control over them. Melt temperature is the actual temperature of the polymer as it exits the nozzle and enters the mold. The barrel setpoints represent the tools we use to arrive at the desired melt temperature, but they are not the same thing.
The mechanical work imparted to the material, the residence time, and the condition of the screw and barrel all play a significant role in determining the actual melt temperature. Similarly, the actual surface temperature of the mold cores and cavities are related to, but not necessarily the same as, the temperature of the fluid passing through the channels in the mold.
Assuming that this is understood, we can examine the effects of these two parameters on the properties of the polymer. It is generally understood that melt temperature has an effect on viscosity. But melt temperature also has an influence on the final molecular weight of the polymer in the molded part.
For example, in an experiment involving parts molded in polypropylene, polymer in parts molded at a melt temperature of 400 F (204 C) had a measurably higher average molecular weight than parts molded at 480 F (249 C). This translated into better impact resistance as well as lower energy consumption in molding and shorter cycle time.
Mold temperature has perhaps a less obvious but often more profound effect on final properties. In amorphous polymers such as ABS and polycarbonate, higher mold temperatures produce lower levels of molded-in stress and consequently better impact resistance, stress-crack resistance, and fatigue performance.
In semi-crystalline materials the mold temperature is an important factor in determining the degree of crystallinity in the polymer. The degree of crystallinity governs many performance parameters, including creep resistance, fatigue resistance, wear resistance, and dimensional stability at elevated temperatures. Crystals can only form at temperatures below the melting point but above the glass-transition temperature (Tg) of the polymer.
Time to Crystallize
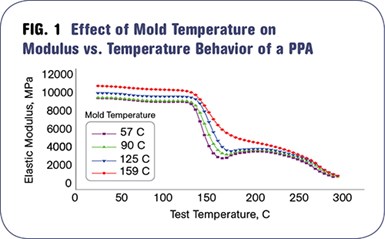
When molding semi-crystalline materials, the ideal mold temperature will be above the Tg in order to give the polymer adequate time to crystallize. Figure 1 compares the behavior of a high-temperature nylon (PPA) when molded at the proper mold temperature and at lower mold temperatures. The plot shows the modulus of the material as a function of temperature. As the mold temperature increases, the stiffness of the material at room temperature also increases.
But the more significant difference between the samples molded at the proper temperature and at the lower mold temperatures can be seen at elevated test temperatures. As the material approaches the glass-transition region at 130 to 140 C, the modulus begins to decline in material molded at lower temperatures and falls farther and faster at even lower mold temperatures. This behavior is in the hands of the processor.
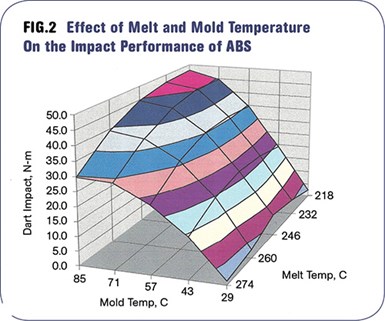
Figure 2 shows the interaction of mold and melt temperatures in determining the impact performance of ABS, an amorphous polymer typically selected for its toughness. The contour plot captures falling-dart impact resistance as the mold temperature is varied from 29 to 85 C (85 to 185 F) and the melt temperature is adjusted from 218 to 271 C (425 to 515 F). It may be surprising to some that the impact resistance ranges from less than 2 N-m (1.4 ft-lb) to almost 50 N-m (36.5 ft-lb) simply as the result of these process changes.
The mold temperature is the dominant factor; however, the best results are obtained when higher mold temperatures are combined with lower melt temperatures. The ideal range of processing conditions, as well as those conditions that should be avoided, are very apparent in this plot.
This behavior is characteristic of all polymers. In general, optimal performance is produced by combining a lower melt temperature with a higher mold temperature. Unfortunately, this is the opposite of what we usually find on the production floor. Typically melt temperatures are running higher than is ideal because melt temperature is often considered to be the only available tool for reducing the melt viscosity. Higher melt temperatures increase energy consumption, degrade the polymer, and extend the cooling time needed to create a dimensionally stable part.
To compensate for this extended cycle time, processors will rely on reduced mold temperatures to gain back lost productivity. Yet a reduced melt temperature combined with a higher mold temperature often produces a part with the same or shorter cycle time and a better set of mechanical properties. When processors understand their role in establishing the properties of the polymer, they approach process development in a very different manner that ultimately reduces cost and improves quality.
ABOUT THE AUTHOR: Michael Sepe is an independent materials and processing consultant based in Sedona, Ariz., with clients throughout North America, Europe, and Asia. He has more than 45 years of experience in the plastics industry and assists clients with material selection, designing for manufacturability, process optimization, troubleshooting, and failure analysis. Contact: (928) 203-0408 • mike@thematerialanalyst.com
Related Content
Melt Flow Rate Testing–Part 1
Though often criticized, MFR is a very good gauge of the relative average molecular weight of the polymer. Since molecular weight (MW) is the driving force behind performance in polymers, it turns out to be a very useful number.
Read MoreThe Effects of Stress on Polymers
Previously we have discussed the effects of temperature and time on the long-term behavior of polymers. Now let's take a look at stress.
Read MorePrices for All Volume Resins Head Down at End of 2023
Flat-to-downward trajectory for at least this month.
Read MoreNew Entrant Heartland Polymers Stepping up as Reliable Supplier
Heartland Polymers’ new Alberta, Canada facility will produce 525 KTA propylene and 525 KTA polypropylene. It is expected to stabilize supply chains across the continent.
Read MoreRead Next
Lead the Conversation, Change the Conversation
Coverage of single-use plastics can be both misleading and demoralizing. Here are 10 tips for changing the perception of the plastics industry at your company and in your community.
Read MoreWhy (and What) You Need to Dry
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreTroubleshooting Screw and Barrel Wear in Extrusion
Extruder screws and barrels will wear over time. If you are seeing a reduction in specific rate and higher discharge temperatures, wear is the likely culprit.
Read More
.jpg;width=70;height=70;mode=crop)