Wood on Plastics: Medical Outlook: All or Nothing?
Medical market filled with opportunities...and risks.
You probably have heard the oft-quoted opening words to the Charles Dickens novel, A Tale of Two Cities: “It was the best of times, it was the worst of times.” But it is the next pair of phrases in this story that might best describe the current state of the medical-device market: “It was the age of wisdom, it was the age of foolishness.”
Never before in the history of mankind have so many people had so much access to such a vast array of medical knowledge and technologies. But because of dysfunctional government policies and mismanagement, the U.S. healthcare system is rapidly approaching a state of crisis. The rate of medical discovery and therapeutic innovation is rapid, but the way the market for these products actually pays for them is convoluted, opaque, and increasingly unbalanced. There is a strong consensus that the market in its current state cannot continue for much longer.
In the near term, the outlook is for steady improvement in the overall market for medical devices. As the economy regains momentum in the coming months, and employment recovers, the demand for medical products will also expand. The global medical devices market is estimated to be more than $300 billion, and the U.S. market accounts for just about a third of that. Both the global and U.S. markets will grow by an annual rate of at least 5% through 2012, which is faster than the expected rate of overall economic growth in the U.S. during the same time period.
So for the next few quarters and years, it is likely that the medical-device business will be good. And there are a number of very strong market fundamentals that suggest business should be good for years to come. The biggest reason is the Baby Boomers. This group represents the largest age cohort in both the U.S. and Europe. They have wealth, and they are just starting to enter their prime medical-device buying age.
The second positive trend (for medical device makers) is the increased incidence of “lifestyle” diseases stemming from issues associated with cardiovascular problems, adult-onset diabetes, hypertension, and obesity. A third positive trend is the new emerging markets that are being created by breakthroughs in the diagnosis and treatment of disease. And finally, as developing economies in Asia and South America acquire new wealth, there are burgeoning populations gaining access to advanced medical care.
All of these reasons combined offer a powerful argument for continued strong growth in demand for medical devices. But so far, the recent rapid rise in demand for these products has been based largely on deficit spending and credit on the part of both federal and state governments.
This situation is rendered even more complicated by President Obama’s healthcare reform initiative.
If left intact, this initiative will create 30 to 40 million potential new consumers of healthcare products through universal coverage. But just what will be covered and whether there will be adequate reimbursement is still a big question. This reform also increases the regulatory pressures on providers and suppliers, and it raises taxes on the medical-device manufacturers.
To say that this has added a great deal of uncertainty to the market for medical devices is an understatement. Over the next decade, there will no doubt be excellent opportunities, but processors supplying this industry should proceed with great caution.
WHAT THIS MEANS TO YOU
•Continue to innovate. Life-saving products will always be more politically acceptable than products that only improve the quality of life.
•Hone up your negotiating skills, as more and more displaced automotive buyers have found positions at medical OEMs.
•Examine growing export opportunities. The Obama Administration has promised to streamline the device-approval process, but new regulations could actually make it more difficult to get some products to market in the U.S.
Related Content
Medical Molder, Moldmaker Embraces Continuous Improvement
True to the adjective in its name, Dynamic Group has been characterized by constant change, activity and progress over its nearly five decades as a medical molder and moldmaker.
Read MoreCatheter Specialist Finds Sweet Spot Serving Small, Medium-Sized Concerns
Medical-component specialist LightningCath has carved a niche meeting the needs of small to medium-sized entrepreneurs with complex catheter designs … quickly.
Read MoreUse Cavity Pressure Measurement to Simplify GMP-Compliant Medical Molding
Cavity-pressure monitoring describes precisely what’s taking place inside the mold, providing a transparent view of the conditions under which a part is created and ensuring conformance with GMP and ISO 13485 in medical injection molding.
Read MoreWisconsin Firms Unite in Battle Against Covid
Teel Plastics opened new plant in record time, partnering with AEC & Aqua Poly Equipment Co. to expand production of swab sticks to fight pandemic.
Read MoreRead Next
Understanding Melting in Single-Screw Extruders
You can better visualize the melting process by “flipping” the observation point so that the barrel appears to be turning clockwise around a stationary screw.
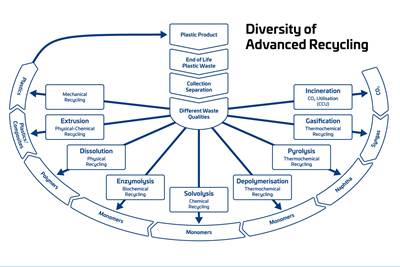
Read MoreAdvanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read More


.png;maxWidth=300;quality=90)