Make Water Treatment Part of Your Process-Cooling Program
Dirty, rusty, smelly water can wreak havoc with even the most meticulous process-cooling program. Here’s how to make sure your water is up to the task.
Do you have a water-treatment program? If so, is it working? Every process application that uses water for heat transfer should be chemically treated.
Chemical treatment will lengthen the life of your equipment and increase operating efficiency. If properly applied and monitored, treatment chemicals do this by protecting metal parts of your system from corrosion and keeping heat-transfer surfaces clean.
Generally speaking, evaporative cooling systems, which usually have a cooling tower or evaporative condenser, are more complex from a water-treatment point of view. Scale, corrosion, and fouling are common to these systems if they lack proper chemical treatment and/or proper water management.
Closed-loop systems, on the other hand, usually do not suffer from mineral scale deposition, since evaporation does not occur in these systems. Corrosion and/or fouling however, are common issues with closed-loop systems. Corrosion can destroy system components or just dissolve metals from one part of the system and redeposit them at another location. The redeposition or fouling often occurs on a heat-transfer surface. Fouling can also cause flow restrictions in critical flow areas.
Here are some simple things to look for that will tell you if you have water-related problems. These tips are directed mostly at processors running closed-loop systems. An example of a closed-loop system is an injection molding application where the molds are cooled or heated by water that is recirculated and not evaporated in a cooling tower or evaporative condenser.
• Do you have compatible metallurgy? Be aware of the different metals in your system. Different metals in direct contact or close proximity to each other—like brass and steel, for example—can cause galvanic corrosion. You can prevent this by insulating the two metals with dielectric couplings.
• Is the water clean or dirty? Clean water does not always mean everything is OK, but dirty water is bad under any circumstance. Rusty red water usually means active corrosion of iron pipe by the oxygen present in the water. Adequate levels of steel corrosion inhibitors will usually keep the water clear. Even if the water looks clear, corrosion product in the form of dissolved iron (ferrous) can still be lurking in solution.
Once it is allowed to set in an open container, the atmospheric oxygen will reduce the clarity of the sample to a red or turbid color.
Black magnetic iron (magnetite) can also discolor the water. It can be there from welding slag, or old rust that has chemically changed. But this is not usually a sign of active corrosion. A blue color in the water is usually a sign of copper or red metal corrosion.
Heat-transfer fluids added to the water often impart a yellow or green color to the water, camouflaging the colors of corrosion cited above. So if you use glycol in your water you’ll have a harder time making these observations. Detecting odor will be more difficult as well.
• Does the water have a peculiar odor? Foul-smelling water is usually an indication of some sort of contamination. A septic, sewer, or gas-type odor can be an indication of microbiological contamination in the water. Bacteria can cause microbiologically influenced corrosion (MIC) of all system metals. Presence of bacteria can easily be tested for. If present, they are usually controlled easily with microbiocides. Once detected they should be monitored regularly. New strains can easily adapt to changing environments.
New construction debris and pipe dope/sealants can cause the water to smell, and should be flushed from the system if detected. These odors usually are constant and do not come and go like the odors from living organisms whose populations fluctuate as the environment changes.
• What’s inside the pipe? Whenever you have the opportunity, you should inspect the system components that are in contact with the water. If a component has failed, cut it open to expose the part that has been in contact with the water. Look for signs of scale or corrosion. Document your observations and record the date the new replacement component was installed. See if improvements made in the water-treatment program coincide with longer life of the replaced component.
Mineral scale is white or tan in color and has a solid, sometimes smooth coating. The most common form of mineral scale, calcium carbonate (CaCO3), fizzes when it comes in contact with a dilute solution of hydrochloric acid. Use a glass container for this test to determine if the mineral is primarily CaCO3
A rusty red deposit may be iron oxide. This may be corrosion from an iron pipe located elsewhere in the system that has redeposited at another location—usually an area of low flow or reduced water velocity. The deposition may have the consistency of mud, or it may be solid and accumulated over time. Copper corrosion product can often impart a sparkly appearance if mixed in with iron.
Corrosion of the system pipe or other metal components is usually obvious to see. Blisters, pits, grooves are all signs of metal corrosion. Pits and grooves are more common to red metals but can occur on any metal.
A slimy coating is usually associated with biofilm. Biofilms are aggregates of predominately bacterial cells attached to and growing on a surface. A biofilm forms when bacteria begin to excrete a slimy, sticky substance that allows them to adhere to surfaces. The biofilm mass often varies with location within a contaminated system, and is typically composed of many species of microorganisms, including bacteria, fungi, algae, and protozoa. All of these conditions are chemically treatable and can be controlled with a dedicated and controlled chemical water treatment program.
All metals have a different pH “comfort zone” or lowest corrosion rate. The metallurgy of your specific system and the critical operating components of your operation will determine the most desirable operating pH for your unique system.
Controlling the pH of a water-cooled injection molding system or any other system is not recommended unless the person doing it has experience as a water-treatment consultant. Overdosing or use of improper testing equipment can result in damage and excessive corrosion of system metals.
Water is locally unique and often requires local knowledge to be treated effectively. Each heating or cooling process that utilizes water for heat transfer offers its own set of challenges. Hire a local water-treatment expert. Often times a local water-treatment professional will be familiar with the challenges local water presents to your process.
One of those challenges is system size. Small systems—less than 5000 gal—do not do require significant amounts of treatment. Often the revenue from small systems is not attractive to chemical-treatment salespeople. They may sell you water-treatment corrosion inhibitors, pH control agents, microbiocides, etc.; but by and large they don’t follow up to see if the operating parameters are being maintained, and there go your results.
When you find a water-treatment consultant to your liking, negotiate a contract where you can specify the frequency of on-site testing included in regular service visits to your facility. You can also ask for corrosion-monitoring equipment to be installed in your system to document the success or failure of specific corrosion inhibitors. Ask your water-treatment consultant for more information on this subject.
Contact the Association of Water Technologies for a local independent water-treatment consultant in your area.
Related Content
Formulating LLDPE/LDPE Blends For Abuse–Resistant Blown Film
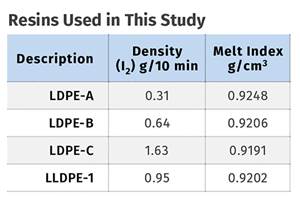
A new study shows how the type and amount of LDPE in blends with LLDPE affect the processing and strength/toughness properties of blown film. Data are shown for both LDPE-rich and LLDPE-rich blends.
Read MoreHow to Select the Right Tool Steel for Mold Cavities
With cavity steel or alloy selection there are many variables that can dictate the best option.
Read MoreTunnel Gates for Mold Designers, Part 1
Of all the gate types, tunnel gates are the most misunderstood. Here’s what you need to know to choose the best design for your application.
Read MoreThe Importance of Melt & Mold Temperature
Molders should realize how significantly process conditions can influence the final properties of the part.
Read MoreRead Next
Understanding Melting in Single-Screw Extruders
You can better visualize the melting process by “flipping” the observation point so that the barrel appears to be turning clockwise around a stationary screw.
Read MoreHow Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read More