MATERIALS: Performance in Polyethylene: Density Matters
In polyethylene, density provides another mechanism to control properties. But it must be specified properly.
In the early part of 2001, processors that mold fuel tanks for small items such as lawn mowers and snowblowers went through a crisis brought about by the removal of a particular grade of polyethylene from the marketplace and lack of an appropriate replacement material. It was an object lesson in a very important principle governing PE material selection: Density matters.
PE is the only commercial polymer that can be selected according to its density. For most polymers density is a constant; polypropylene is 0.90 g/cm3, polycarbonate is 1.2 g/cm3, etc. Adding filler such as talc or glass will increase the density; incorporating an impact modifier may lower it slightly; but the density of the base polymer does not change.
PE, however, can be manufactured across a range of densities that are related to the spacing between the polymer chains. The original PE that was created in the 1930s by placing ethylene gas under extremely high pressures and temperatures was what today is called low-density PE (LDPE). It was not called LDPE when it was first developed, because there was no other kind. It was simply polyethylene. It was soft, flexible, and resistant to stress cracking; a perfect replacement for the crosslinked rubber materials that were then being used for wire insulation.
In the mid-1950s, new catalyst technologies enabled polymer chemists to control the way that the polyethylene chain grew during the polymerization process. The polymer chains in LDPE contained numerous branches of varying lengths that created substantial amounts of empty space between polymer chains. The new catalysts produced chains that were comparatively free of these side branches, resulting in a linear structure that allowed the molecules to pack more closely together. This closer packing increased the intermolecular forces between the chains, resulting in a higher density material that was noticeably stronger, stiffer, and more creep resistant. It was also less ductile and more susceptible to stress cracking, especially at low temperatures.
As an interesting aside, the same catalysts that made HDPE possible also gave polymer chemists a new level of control over other types of molecular arrangements. This turned polypropylene from an adhesive to a semi-crystalline polymer with mechanical and thermal properties that were superior in many ways to those of polyethylene.
With increasing control over the polymerization reaction, chemists were able to vary the density of PE on an essentially continuous scale from as low as 0.912 g/cm3 to as high as 0.975 g/cm3. A classification system grew up to categorize the different families according to their density. Low-density, medium-density, high-density copolymers, and high-density homopolymers became known as Type I, Type II, Type III, and Type IV, respectively. As polymers made an increasing impact on various industries, engineers learned how to select materials for various applications based on the balance of strength, stiffness, and toughness that each class of polyethylene displayed. Even within a given type class, there was a significant density range of approximately 0.014 to 0.017 g/cm3 that separated the highest and lowest density materials.
In the early 1980s another catalyst breakthrough made it possible to control the length and frequency of the branches in the lower density range, giving rise to linear low-density PE (LLDPE). While this provided an improved set of properties in this lower density range, it did not change the available density range. Then in the early 1990s the metallocene or single-site catalysts changed all that. Suddenly it was possible to control the molecular structure of the polymer at an unprecedented level. It also became possible to produce polyethylene materials with densities below that of polypropylene; all the way down to 0.857 g/cm3. These materials were very soft and flexible, had very low melting points, and created a whole new set of performance profiles for the neat resins as well as for products that used these new materials as impact modifiers.
Because the density of PE can be controlled and varied, it must be specified as part of the material selection process. If you talk to people who spend a lot of time around PE, they tend to talk in terms of two numbers that tell them a great deal about the material they are using. A material may be described in the industry vernacular as a “7-melt 953.” This is a shorthand reference to two very important properties of the material: the melt index or melt flow rate, which is a relative indicator of the average molecular weight of the material; and the density, which is related to an extensive range of thermal and mechanical properties. The 953 has a decimal point in front of it, as in 0.953 g/cm3. Increasingly you may also hear one more adjective in the shorthand description—the term “narrow” or broad.” This refers to the molecular-weight distribution of the material, but that is for another article.
PE has been a fixture in the world of commercial plastics for such a long time that it is tempting to think of it as a simple material. After all, it is a commodity, consumed worldwide at a rate of tens of billions of pounds each year in applications that often seem rather mundane. But consider the intricacies of selecting a material where variations in density are possible. The 7-melt polymer with a density of 0.953 g/cm3 is a prime candidate for molding pails. One-, two-, and five-gallon PE pails have been around for nearly 40 years. They embody many of the competing objectives and compromises that go into the material selection process.
Pails, once filled with their contents, may be stacked so that the bottom pail in the stack is under a considerable constant load. In the 5-gal variety, each pail may hold 40-60 lb of stuff. Creep resistance is therefore an important requirement. If the storage conditions involve elevated temperatures, the degree of difficulty in fulfilling this requirement increases.
At the same time, the full pail may be dropped, creating an impact load. Toughness and creep resistance are competing objectives; improving one will typically reduce the other one. Over time, the combination of molecular weight and density in “pail” materials has been fine-tuned to strike the optimal balance between these competing properties. If it falls too far below the target, the material loses strength and stiffness and the applied load causes the stack of pails to collapse. If the density increases, the material becomes less ductile and full pails will crack when dropped.
In most polymers, you can improve impact performance by selecting a higher molecular-weight material grade without detracting from the rest of the property profile. This is where the melt flow rate comes in, a topic we discussed at length in a previous series of articles. But molecular weight cannot be increased indefinitely without challenging the processor. In PE, density provides another mechanism to control properties. But it must be specified properly.
Often the window of appropriate densities for a given application is quite small. And this is exactly the problem that the fuel tank manufacturers ran into. Next month we will review the apparently small but critical change that was made to the material specification for this product and the far-reaching consequences it had for everyone involved.
Related Content
Melt Flow Rate Testing–Part 1
Though often criticized, MFR is a very good gauge of the relative average molecular weight of the polymer. Since molecular weight (MW) is the driving force behind performance in polymers, it turns out to be a very useful number.
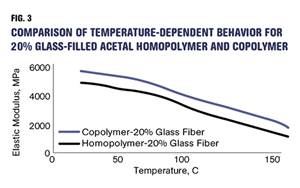
Read MoreHow Do You Like Your Acetal: Homopolymer or Copolymer?
Acetal materials have been a commercial option for more than 50 years.
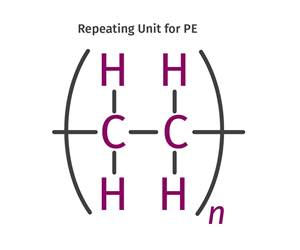
Read MoreThe Fundamentals of Polyethylene – Part 2: Density and Molecular Weight
PE properties can be adjusted either by changing the molecular weight or by altering the density. While this increases the possible combinations of properties, it also requires that the specification for the material be precise.
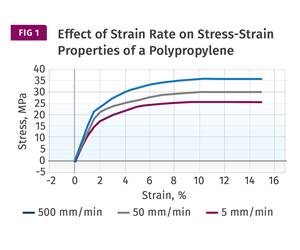
Read MoreUnderstanding Strain-Rate Sensitivity In Polymers
Material behavior is fundamentally determined by the equivalence of time and temperature. But that principle tends to be lost on processors and designers. Here’s some guidance.
Read MoreRead Next
Understanding Melting in Single-Screw Extruders
You can better visualize the melting process by “flipping” the observation point so that the barrel appears to be turning clockwise around a stationary screw.
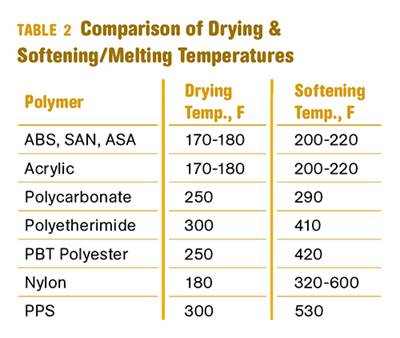
Read MoreWhy (and What) You Need to Dry
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read More.jpg;width=70;height=70;mode=crop)