Part 11: A Processor's Most Important Job
It’s the processors job to ensure molded parts contain enough stabilizer to perform to the expectations of the end use.
The final aspect of processing that is designed to ensure good retention of mechanical properties involves taking good care of the additive package. All commercial polymers contain an additive package, a combination of compounds designed to provide the stability needed to allow melt processing to occur without degrading the polymer. Resins may also contain additives provided to enhance end-use performance. The constituents in the additive package can vary from compound to compound, and may include UV stabilizers, nucleating agents, antistatic agents, antimicrobials, colorants and flame retardants. But at minimum they are likely to include some level of lubrication and some degree of stabilization against thermal and oxidative degradation. It is this stabilizer package that we are most interested in for purposes of this discussion.
Antioxidants, as these compounds are generally referred to, are incorporated in a material for two purposes: to allow the material to survive the high temperatures and shear stresses of the molding process; and to provide stability that may be needed so that the part can fulfill its application requirements. Often these functions are delegated to two different sets of chemicals, classified as primary and secondary antioxidants. The chemistry of these materials is fascinating and complex. But when all is said and done, the purpose of these substances is to protect the polymer from degradation that can occur during processing and in use.
The exact amounts of these additives will depend upon the polymer being stabilized and the applications that the material is intended for. Polypropylene and polyethylene are good examples of materials that require protection from oxidation. Generally, PE is more resistant to oxidation than PP and therefore requires lower loadings of the additives. But all commercial PE and PP materials require some minimum level just to get through melt processing. If after the part is molded it is expected to withstand elevated temperatures while in application, a higher level of stabilization will be needed to ensure that the product does not fail. For PP, these types of high-temperature applications can be anything from coffee-maker components to tanks for holding automobile engine coolant.
The material supplier will incorporate a certain amount of the stabilization package in the material, making this the starting point for that material. It is now the processor’s job to handle the material during processing so that the molded part contains enough stabilizer to perform to the expectations of the end user. The molding process will always consume some amount of the antioxidant in the material. This is expected. But the amount of stabilizer that is consumed will depend upon the process conditions, most notably the melt temperature and the time the material spends in the molten state. Lower melt temperatures and shorter residence times will produce parts that retain a higher level of stabilization, and these parts will be more capable of handling the application environment. If melt temperatures become elevated or residence times become extended, the molded part will be less capable of managing the application demands.
In the lab we can measure the stability of a material against oxidation. Using an analytical technique known as differential scanning calorimetry (DSC), we can expose a sample of material to a constant elevated temperature in the presence of oxygen and determine the time it takes for the material to undergo oxidation. The measurement is known as the oxidation induction time (OIT) and the technique was first used to help polymer scientists understand the premature failure of wire insulation in the 1960s. The accompanying graph shows a result from this test. The sudden change in the slope of the plot is associated with the onset of oxidation. This raw material can withstand 77 min of exposure at these conditions before the antioxidant package is consumed and the material rapidly fails.
If this test is run on parts molded from this material, the result can be used to determine how the process influenced the long-term stability of the material. In practice, we have observed molded parts that retain as much as 75% of the OIT of the raw material. and we have seen parts that preserve as little as 10% of this baseline value. The difference between these two outcomes is related to the thermal history of the process. And the lifetime of the part in the field will be governed to a significant extent by the level of oxidative stability that the part has when it goes into use.
This has implications for the use of regrind. Regrind can be expected to exhibit some level of depletion in the antioxidant package. The degree to which this occurs will depend upon the process conditions. Often, I am asked how much regrind should be used or how many times a material can undergo melt processing. The answer is always that it depends upon the thermal stresses the material was exposed to when it passed through the molding process previously. This depletion can also occur while the part is in use.
Recently, I reviewed an analysis of a material that had an OIT of 140 min in pellet form. The as-molded parts retained an OIT of 100 min, and parts that had been exposed to a prolonged elevated temperature routine were at 60-70 min. As we move in the direction of using more post-consumer recycled material, it will become increasingly important that we consider the life cycle of the initial product as well as the types of products that we plan to make from the recycled material. It may be necessary to reconstitute the stabilizers when producing the regenerated raw material.
It has taken us some time to review all the aspects of processing that relate to maintaining the integrity of the polymer. All of these are factors that are not part of most quality plans associated with molding parts. Processors are typically focused on making parts to print at a cycle time that allows them to make a profit. The molecular weight, polymer structure, level of internal stress, and integrity of the reinforcements and additives are seldom considered to be part of the job.
Many processors may not even be aware of their role in achieving optimal results in these areas. There are some interesting common strategies in doing so. For example, molecular weight and additive retention are both optimized by running materials at the lower end of the melt-temperature range. Maximizing crystallinity and minimizing molded-in stress are both achieved by running higher mold temperatures. Once the processor becomes aware of their role in these matters, they can use this understanding to inform their process-development strategies. The world of plastic products will be a better place for having gone through the learning process.
ABOUT THE AUTHOR: Mike Sepe is an independent, global materials and processing consultant whose company, Michael P. Sepe, LLC, is based in Sedona, Ariz. He has more than 40 years of experience in the plastics industry and assists clients with material selection, designing for manufacturability, process optimization, troubleshooting, and failure analysis. Contact: (928) 203-0408 • mike@thematerialanalyst.com.
Related Content
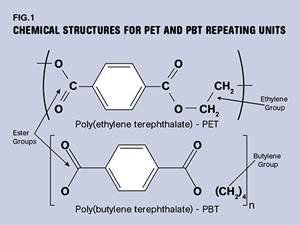
PBT and PET Polyester: The Difference Crystallinity Makes
To properly understand the differences in performance between PET and PBT we need to compare apples to apples—the semi-crystalline forms of each polymer.
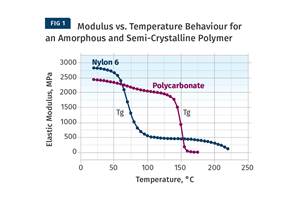
Read MoreThe Effects of Temperature
The polymers we work with follow the same principles as the body: the hotter the environment becomes, the less performance we can expect.
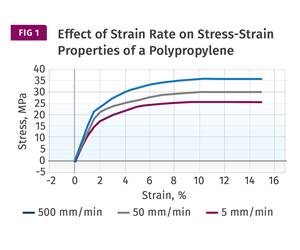
Read MoreUnderstanding Strain-Rate Sensitivity In Polymers
Material behavior is fundamentally determined by the equivalence of time and temperature. But that principle tends to be lost on processors and designers. Here’s some guidance.
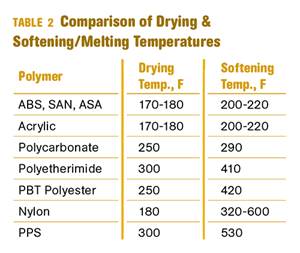
Read MoreWhy (and What) You Need to Dry
Other than polyolefins, almost every other polymer exhibits some level of polarity and therefore can absorb a certain amount of moisture from the atmosphere. Here’s a look at some of these materials, and what needs to be done to dry them.
Read MoreRead Next
Advanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read MoreTroubleshooting Screw and Barrel Wear in Extrusion
Extruder screws and barrels will wear over time. If you are seeing a reduction in specific rate and higher discharge temperatures, wear is the likely culprit.
Read MoreHow Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read More
.jpg;width=70;height=70;mode=crop)



















.png;maxWidth=300;quality=90)