Try This Approach to Clean Up Your Recycled Material
CO2 can be used as a totally “green” solvent to remove many kinds of contamination in recycled plastics without the need for toxic solvents.
Recycling is getting lots of attention these days due to public and media outcries about plastic in the environment. About half of all the plastics produced go into single-service food packaging of some type. Food packaging usually contains various oils, printed surfaces, surface treatments and adhesives that are difficult to remove, even by very aggressive washing, and require the use of toxic solvents that often make the reclaim unusable for further food-packaging applications. This makes it hard for processors running reclaim to produce the clean products the application may demand.
However, supercritical CO2 can be used as a totally “green” solvent to remove many kinds of contamination in recycled plastics without need for toxic solvents. CO2 is naturally occurring in the atmosphere, and when stripped of residual contamination after processing it can either be reclaimed or simply vented back into the atmosphere. The use of supercritical CO2 is a well-established technology in food and drug processing. For many years it has been used to decaffeinate coffee and tea, for example.
CO2 exists naturally as a gas in the atmosphere; at reduced temperatures it is a solid (dry ice). However under certain conditions CO2 can exist as a liquid if the pressure and temperature are increased above a certain point. The phase diagram shows the pressures and temperatures necessary for CO2 to exist in the liquid phase.
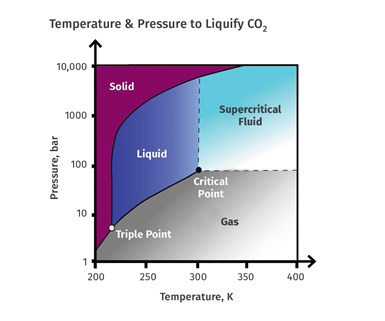
Shown here are the pressures and temperatures necessary for CO2 to exist in the liquid phase.At the tripe point all three phases can exist, but as the temperature and pressure are increased the CO2 becomes a liquid. SOurce: Wikipediaons CO2 can exist as a liquid if the pressure and temperature are increased above a certain point. The phase diagram shows the pressures and temperatures necessary for CO2 to exist in the liquid phase.
At the triple point (TP) all three phases can exist, but as the temperature and pressure are increased the CO2 becomes a liquid. At a pressure of 1070 psi and 89.8 F the liquid enters a supercritical phase (CP) and remains in that phase with increasing pressure and temperature. That is important because in that phase it has some unusual properties—between a gas and a liquid—that allow it to more easily penetrate through many solids (like polymers) and act as a strong solvent to dissolve many materials. Its solvent properties are so strong, in fact, that when the supercritical phase exists it must be contained in highly corrosion-resistant materials.
CO2 treatment has very little effect on the polymer morphology, making the recycled polymer useful for the original performance requirements.
Although the technology of decontamination with supercritical CO2 is old and well understood, this is typically a batch process because of the difficulty in controlling pressure from the treatment phase, where the solvent (CO2) absorbs the contaminants, to the solvent-cleaning phase, where the pressure is reduced and the contaminants drop out of solution. If the pressure is released during the treatment phase, the residual contamination simply drops out of the supercritical CO2 and right back into the polymer. The use of an extruder simplifies that by allowing the pressures to be easily segregated by use of viscous seals between the treatment and solvent-cleaning phase.
The use of a viscous seal before and after the area to be cleaned by the supercritical CO2 isolates the “treatment area” so that the pressure and temperature necessary to maintain the CO2 in the supercritical condition can easily be maintained without elaborate pressure chambers and transfer valves. The CO2 can be simply exhausted into a containment container at a lower pressure, allowing the residuals to drop out, and then return to the atmosphere with no net environmental effect. However, in the interest of economics the “clean” CO2 can be recompressed and reused.
Fluropolymers are the only common polymers that are dissolved by the supercritical CO2 so this treatment has broad applicability. Amorphous polymers seem to be more effectively cleaned using this technique, but crystalline polymers apparently can be cleaned effectively with longer treatment times and additional mixing in the treatment section.
The CO2 treatment has very little effect on the polymer morphology, making the recycled polymer useful for the original performance requirements. Although somewhat of a change from typical decontamination processes used in recycling, it offers the potential to greatly expand the amount of recycle that can be reused for its original purposes.
About the Author: Jim Frankland is a mechanical engineer who has been involved in all types of extrusion processing for more than 40 years. He is now president of Frankland Plastics Consulting, LLC. Contact jim.frankland@comcast.net or (724) 651-9196.
Related Content
Advanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read MoreCompatibilizers Aid Recycling & Upcycling of Mixed Resins
Compatibilizers are proving their worth in boosting critical properties such as impact/stiffness balance of PCR and PIR blends of polyolefins and other plastics.
Read MoreSD Polymers: 'One-Stop Solution for Mechanical Recycling'
‘Passionate’ recycler invests in people and technology to meet commitment to innovative, sustainable solutions for its processor customers.
Read MoreScaling Up Sustainable Solutions for Fiber Reinforced Composite Materials
Oak Ridge National Laboratory's Sustainable Manufacturing Technologies Group helps industrial partners tackle the sustainability challenges presented by fiber-reinforced composite materials.
Read MoreRead Next
Troubleshooting Screw and Barrel Wear in Extrusion
Extruder screws and barrels will wear over time. If you are seeing a reduction in specific rate and higher discharge temperatures, wear is the likely culprit.
Read MoreAdvanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read MorePeople 4.0 – How to Get Buy-In from Your Staff for Industry 4.0 Systems
Implementing a production monitoring system as the foundation of a ‘smart factory’ is about integrating people with new technology as much as it is about integrating machines and computers. Here are tips from a company that has gone through the process.
Read More
.jpg;width=70;height=70;mode=crop)













.png;maxWidth=300;quality=90)