Materials: Annealing Tips for Amorphous Polymers, Part 2
In amorphous polymers, annealing is performed to draw down the internal stresses to a level not achievable within the conditions of a normal molding process. But a few parameters are important to achieving the desired results.
As we discussed briefly in Part 1 last month, amorphous polymers are susceptible to failure by environmental stress cracking (ESC). We understand this mechanism to be essentially a mechanical failure that is accelerated by the presence of a chemical that locally plasticizes the polymer in an area where a small defect has been created.
The defect can be an inclusion such as a piece of metal or carbon char or it can be a notch created by incidental damage. It may also be due to a design defect such as a sharp corner or a rapid change in the wall thickness of the part that creates a locally elevated stress level. Or it can be promoted by an elevated level of internal stress caused by molding conditions. High levels of internal stress are caused by rapid cooling of the polymer.
A processing strategy that involves rapid cooling can also influence short-term properties, most notably ductility. This is a concern since many amorphous polymers, such as ABS and PC, are used in large part because of their excellent toughness. Figure 1 shows the results of a study on the effects of melt and mold temperature on the impact resistance of ABS. This shows that the molded specimens exhibit very low energy to break when the mold temperature is set relatively low. As the mold temperature is increased the impact resistance rises dramatically.
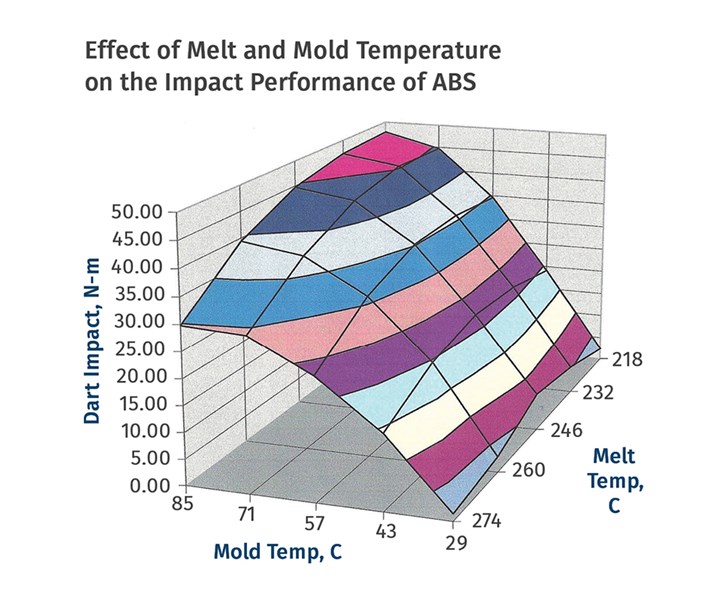
FIG 1 Molded specimens exhibit very low energy-to-break when the mold temperature is set relatively low. As the mold temperature is increased the impact resistance rises dramatically.
But even with a high mold temperature, the cooling rate of a polymer during the injection molding process is on the order of 150-300° C/min (270-540° F/min). With such a rapid change in temperature, some level of internal stress is inevitable. In situations where the application environment involves some combination of elevated temperature, extended lifetime, stresses that may exceed the proportional limit, and exposure to certain chemicals, even relatively low levels of internal stress may result in premature failure due to ESC. Failure-analysis meta studies have shown that ESC is the leading cause of field failures in plastic parts and this failure mode primarily affects amorphous polymers.
In amorphous polymers, annealing is performed to draw down the internal stresses to a level not achievable within the conditions of a normal molding process. There are a few parameters that are important to achieving the desired results. The first of these is the temperature of the annealing process. Typically, the recommended annealing temperature is indexed to the glass-transition temperature (Tg) of the polymer. This can be readily measured by analytical techniques such as differential scanning calorimetry (DSC) or dynamic mechanical analysis (DMA). DMA has the advantage of measuring the physical properties of the polymer, therefore it provides more information about the range of temperatures that can be used to relax the internal stresses in the part.
Failure-analysis meta studies have shown that ESC is the leading cause of field failures in plastic parts and this failure mode primarily affects amorphous polymers.
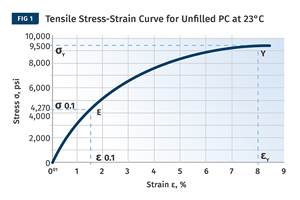
Figure 2 provides a plot of the elastic modulus as a function of temperature for a typical PC. The Tg occurs in the temperature region where the elastic modulus of the polymer declines rapidly over a very narrow temperature range of 140-155 C (284-311 F).
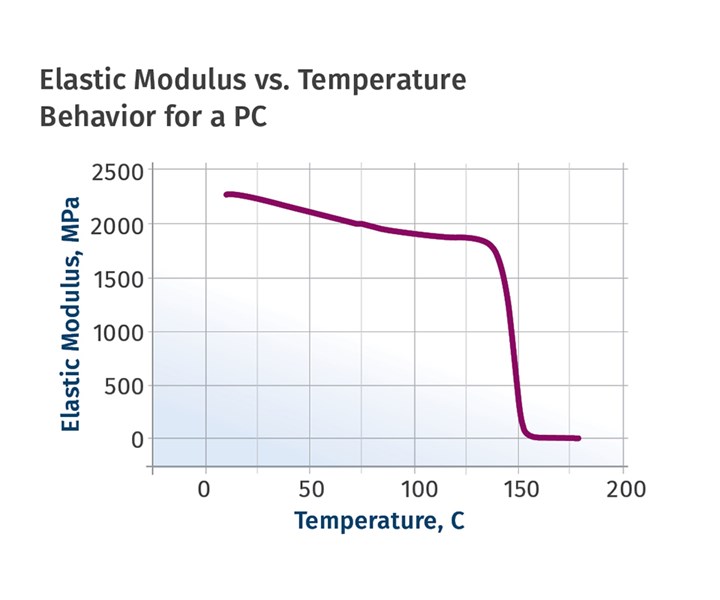
FIG 2 The glass-transition temperature occurs in the temperature region where the elastic modulus of the polymer declines rapidly over a very narrow temperature range of 140-155 C (284-311 F).
Recommendations of an appropriate annealing temperature for polycarbonate vary between 121 C (250 F) and 135 C (275 F). These temperatures are close to the Tg but remain below the onset of the rapid decline in modulus in order to prevent deformation of the parts. The objective is to use a temperature as close to this onset as possible without producing part distortion or an excessive degree of dimensional change. This will depend somewhat on part geometry and the level of support that can be provided to areas that tend to be most susceptible to distortion, such as areas around gates.
The second important parameter is the annealing time. This will depend upon the part thickness. Plastics are relatively poor conductors of heat, and the part must be allowed to come to a uniform temperature throughout. Typical recommendations are a minimum of 30 min once the parts have achieved the desired temperature, plus 5 min/mm (0.040 in.) of wall thickness. For parts with sections thicker than 6 mm (0.250 in) the best results are obtained by doubling this time. Failure to provide sufficient time to reach and maintain a uniform temperature for an appropriate amount of time may actually produce an increase in the level of internal stress.
Perhaps the most important condition associated with annealing is the rate of temperature change, particularly the rate of change that occurs during the cooling process. Ideally, the parts should be heated from room temperature to the annealing temperature at a rate of no more than 50° C/hr (90° F/hr). But it is the cooldown portion of the annealing process that has the greatest influence on the result. Here again, specific recommendations vary.
However, a good guideline is a cooling rate no faster than 25 C/hr (45 F/hr) until the parts have reached a temperature of 60-65 C (140-149 F). Some parts may need to be cooled at a rate as slow as 5° C/hr (9° F/hr). The most common mistake that results in an unsatisfactory annealing outcome is cooling too rapidly. Often parts are removed from the oven as soon as the prescribed annealing time is completed. The parts cool rapidly from the annealing temperature to room temperature, undoing all the work that was done by the annealing process.
A good guideline is a cooling rate no faster than 45° F/hr until the parts have reached a temperature of 140-149 F.
The ultimate test of the efficacy of an annealing process is a solvent stress-crack evaluation. For each polymer there is a chemical or a mixture of chemicals that will target a certain threshold of internal stress. Often this approach involves a mixture of two substances. One acts as the inert ingredient while the other is the active ingredient that promotes stress cracking. By changing the ratio of these two constituents in the mixture, the targeted threshold stress can be adjusted so that the stress in the part can be measured precisely.
ABS, for example, uses a mixture of an acetate such as ethyl acetate and an alcohol such as ethanol. Higher concentrations of the acetate required to induce stress cracking correlate to lower internal stresses in the part. The same approach is used in polycarbonate. However, with polycarbonate the mixture is one of n-propanol and toluene. Parts are immersed in the mixture for a prescribed amount of time, removed and rinsed, and then evaluated for cracking. The location of any observed cracking helps to identify areas of the part that are susceptible to the formation of elevated levels of stress.
An alternative approach uses a single reagent and the immersion time required to produce stress cracking is related to the internal stress in the part. As an example, polycarbonate can be tested using propylene carbonate. The level of internal stress in the part is a function of the time that the part is immersed in the fluid. With either method, an effective annealing process will produce a notable reduction in the measured threshold stress.
Annealing of semi-crystalline polymers is done for a completely different reason. In our next segment we will discuss this process and the guidelines for getting most out of annealing this class of polymers.
ABOUT THE AUTHOR Mike Sepe is an independent, global materials and processing consultant whose company, Michael P. Sepe, LLC, is based in Sedona, Ariz. He has more than 40 years of experience in the plastics industry and assists clients with material selection, designing for manufacturability, process optimization, troubleshooting, and failure analysis. Contact: (928) 203-0408 • mike@thematerialanalyst.com.
Related Content
The Effects of Stress on Polymers
Previously we have discussed the effects of temperature and time on the long-term behavior of polymers. Now let's take a look at stress.
Read MoreNew Entrant Heartland Polymers Stepping up as Reliable Supplier
Heartland Polymers’ new Alberta, Canada facility will produce 525 KTA propylene and 525 KTA polypropylene. It is expected to stabilize supply chains across the continent.
Read MoreThe Strain Rate Effect
The rate of loading for a plastic material is a key component of how we perceive its performance.
Read MoreUnderstanding the ‘Science’ of Color
And as with all sciences, there are fundamentals that must be considered to do color right. Here’s a helpful start.
Read MoreRead Next
Materials Part 1: What Annealing Can Do for Your Process
Relatively rapid cooling rates in processing introduce internal stress. If functional problems in use result, annealing may draw down the stress to levels that may not be achievable during processing.
Read MoreHow Polymer Melts in Single-Screw Extruders
Understanding how polymer melts in a single-screw extruder could help you optimize your screw design to eliminate defect-causing solid polymer fragments.
Read MoreAdvanced Recycling: Beyond Pyrolysis
Consumer-product brand owners increasingly see advanced chemical recycling as a necessary complement to mechanical recycling if they are to meet ambitious goals for a circular economy in the next decade. Dozens of technology providers are developing new technologies to overcome the limitations of existing pyrolysis methods and to commercialize various alternative approaches to chemical recycling of plastics.
Read More
.jpg;width=70;height=70;mode=crop)







.png;maxWidth=300;quality=90)